Neutral electrolyzed saline vs Standard care (RCT)
Mild outpatients
FOREST PLOTS -2022-04-29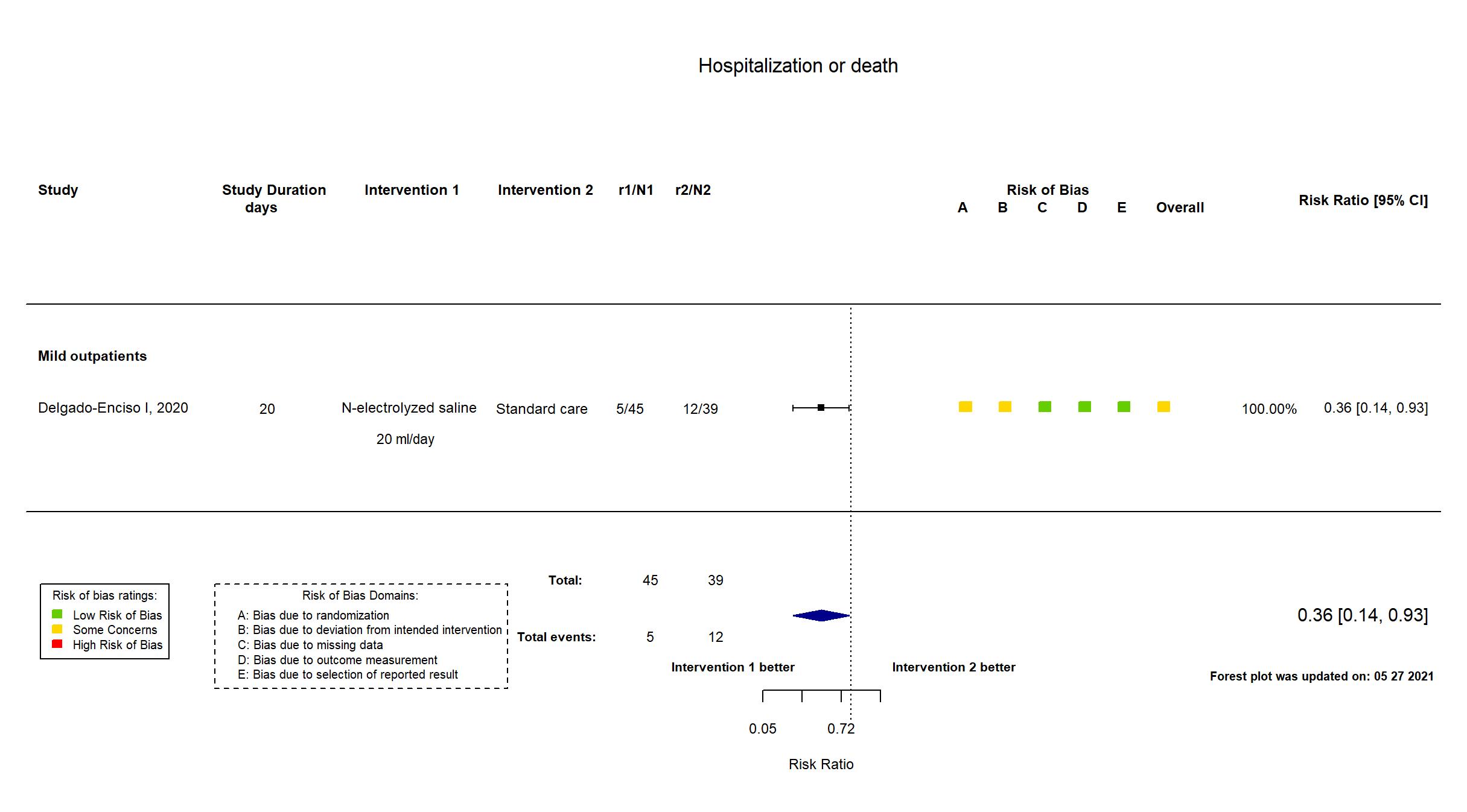



Trial RPCEC00000309
Publication Delgado-Enciso I, Exp Ther Med (2021) (published paper)
Dates: 2020-05-01 to 2020-06-30
Funding: Mixed (Consejo Estatal de Ciencia y Tecnología del Estado de Colima; Esteripharma S.A. de C.V.)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: | |
| Location :
Multicenter / Mexico Follow-up duration (days): 20 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
N-electrolyzed saline 5 mL nebulization 4 times a day for 10 days. IV injections with different doses and frequencies could also be administered. |
|
| Control
Standard care | |
| Participants | |
| Randomized NR Analyzed 214 participants N-electrolyzed saline =110 Standard care=104 | |
| Characteristics of participants N= 214 Mean age : NR 108 males Severity : Mild: n= 214/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Disease progression (hospitalization or deaths) until 14th day. | |
| In the report Number of patients with disease progression (hospitalization or death); Patient Acceptable Symptom State (PASS); change from the baseline in the patient overall self-assessment (score 0-10) | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication:
|
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to available version of the journal and pre-print articles, the study registry was used in data extraction. The study achieved the target sample size (n=100) specified in the registry. There was no change from the trial registration in the intervention and control treatments. Some co-primary outcomes detailed in the report were not specified in the registry (Patient Acceptable Symptom State (PASS) and the change from the baseline in the
patient overall self-assessment). The same applies to a secondary outcome stated in the report that was not in the registry, such as changes from the baseline in different types of body pain (arthralgia, myalgia, headache, and sore
throat), or more exactly, the difference between the values at enrollment and on days 3, 5, and 7 of follow-up.
Pain was measured on the 0–10 VAS.
This is the report of data on the effect of the experimental treatment on “Patients treated at home”. Adverse events and viral negative conversion events among the control group were not reported. This trial was updated on September 29th, 2021 with data from the peer-reviewed journal publication. |