Hyperimmune anti-COVID-19 Intravenous Immunoglobulin vs Standard care/Placebo (RCT)
Mild outpatients
FOREST PLOTS -2022-12-15
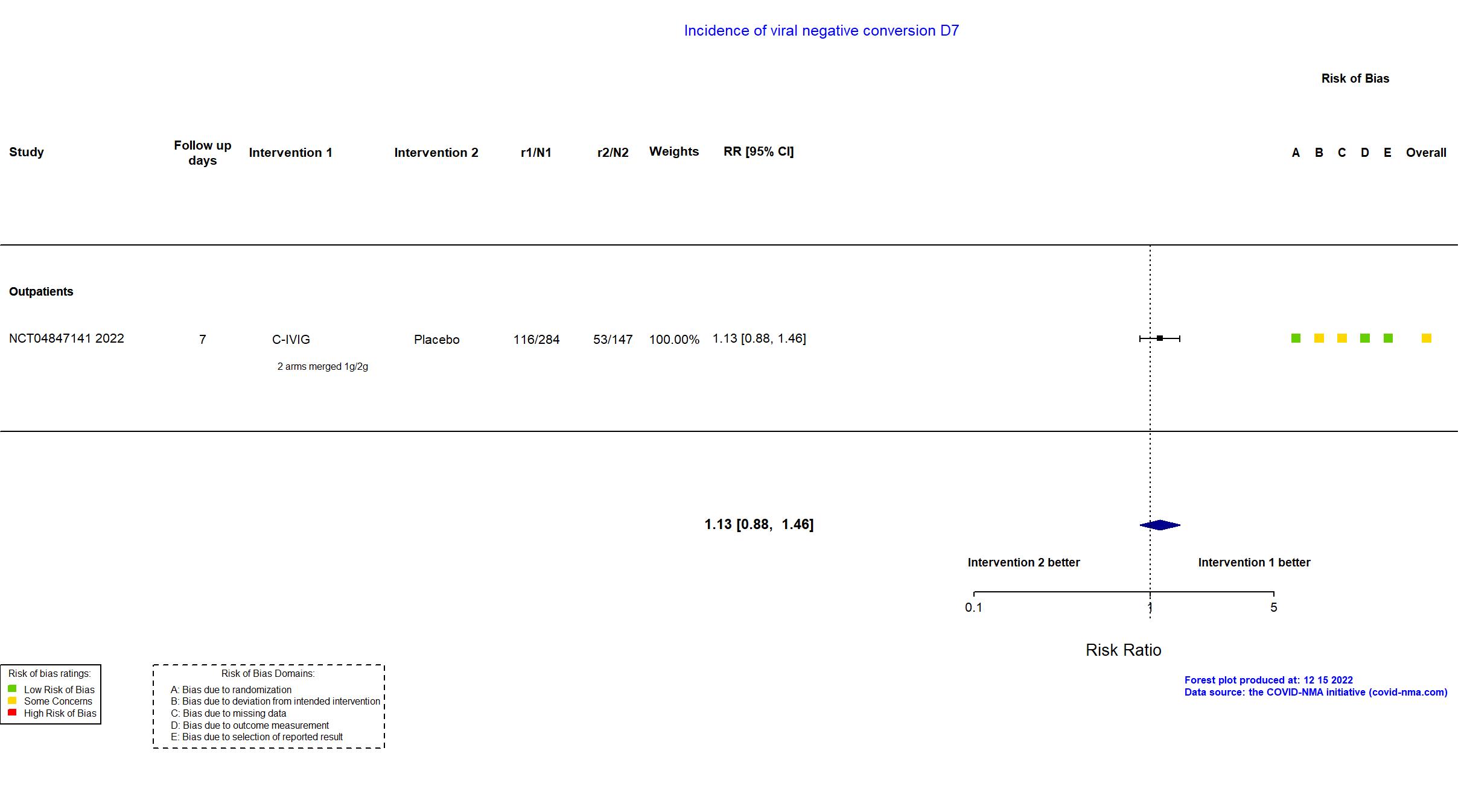
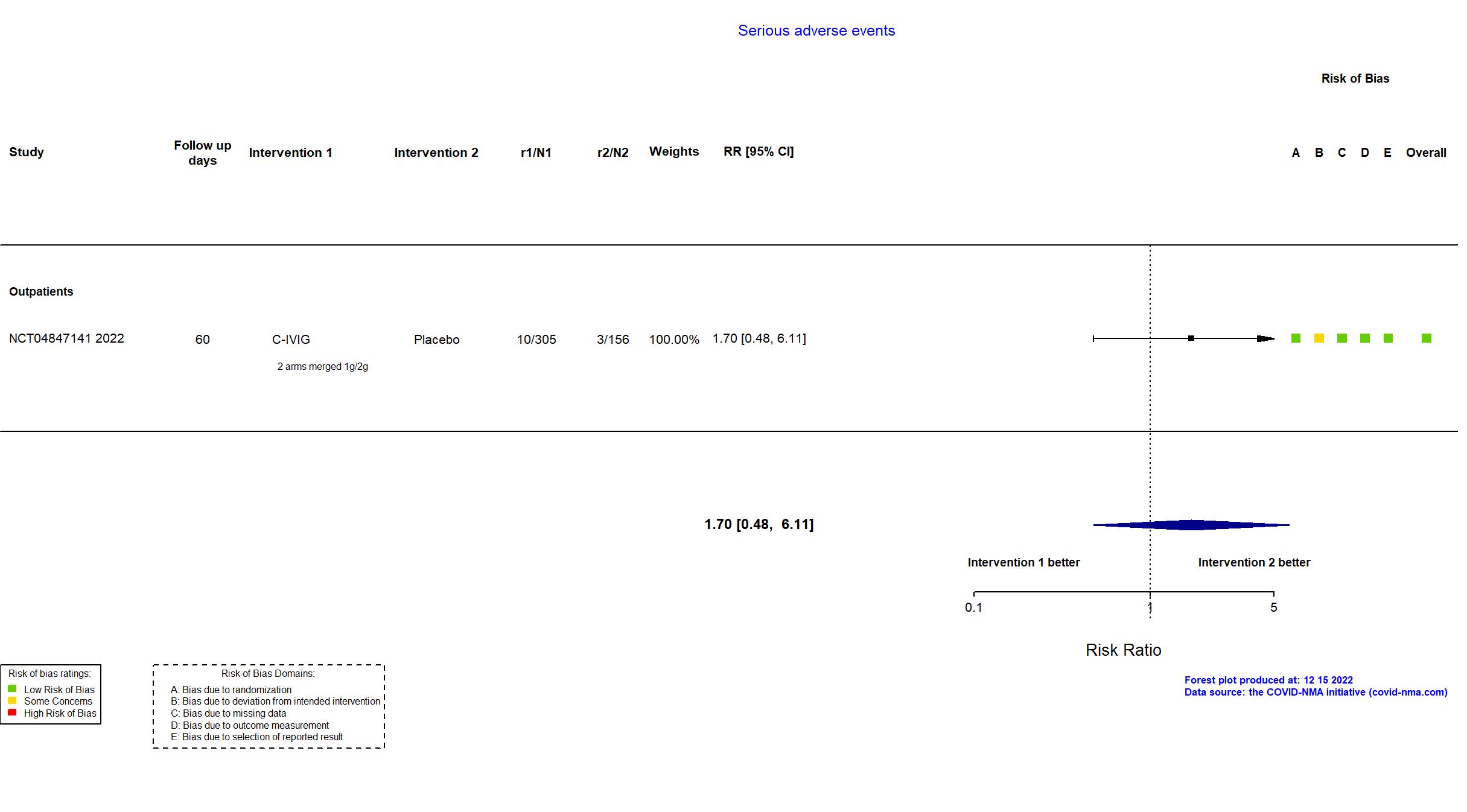






Trial NCT04847141
Publication NCT04847141, Unpublished (2022) (results posted on registry)
Funding: Not reported/unclear
Conflict of interest: *
| Methods | |
| RCT Blinding: quadruple blinding | |
| Location :
Multicenter / Spain Follow-up duration (days): 60 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
C-IVIG 2g 2g subcutaneously single dose C-IVIG 1g 1g subcutaneously single dose C-IVIG 1g subcutaneously single dose |
|
| Control
Placebo | |
| Participants | |
| Randomized NR Analyzed 766 participants Placebo=156 C-IVIG 2g=153 C-IVIG 1g=152 C-IVIG=305 | |
| Characteristics of participants N= 766 Mean age : NR 350 males Severity : Mild: n= 0/ Asymptomatic: n=613 | |
| Primary outcome | |
| In the register Percentage of Asymptomatic Participants Who Remained Asymptomatic, i.e., Who Did Not Develop Symptomatic COVID-19 Through Day 14 [ Time Frame: Up to Day 14 ] | |
| In the report NR | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | This is an unpublished trial whose results have been reported in ClinicalTrials.gov. The trial registry, protocol and statistical analysis plan were used in data extraction and assessment of risk of bias. The trial was registered prospectively and no important changes were made to primary or secondary outcomes after recruitment start. The trial (n = 465) did not achieve its target sample size (n = 801). The study was terminated for futility.. |