Nitazoxanide vs Standard care/Placebo (RCT)
Mild outpatients
FOREST PLOTS -2022-08-18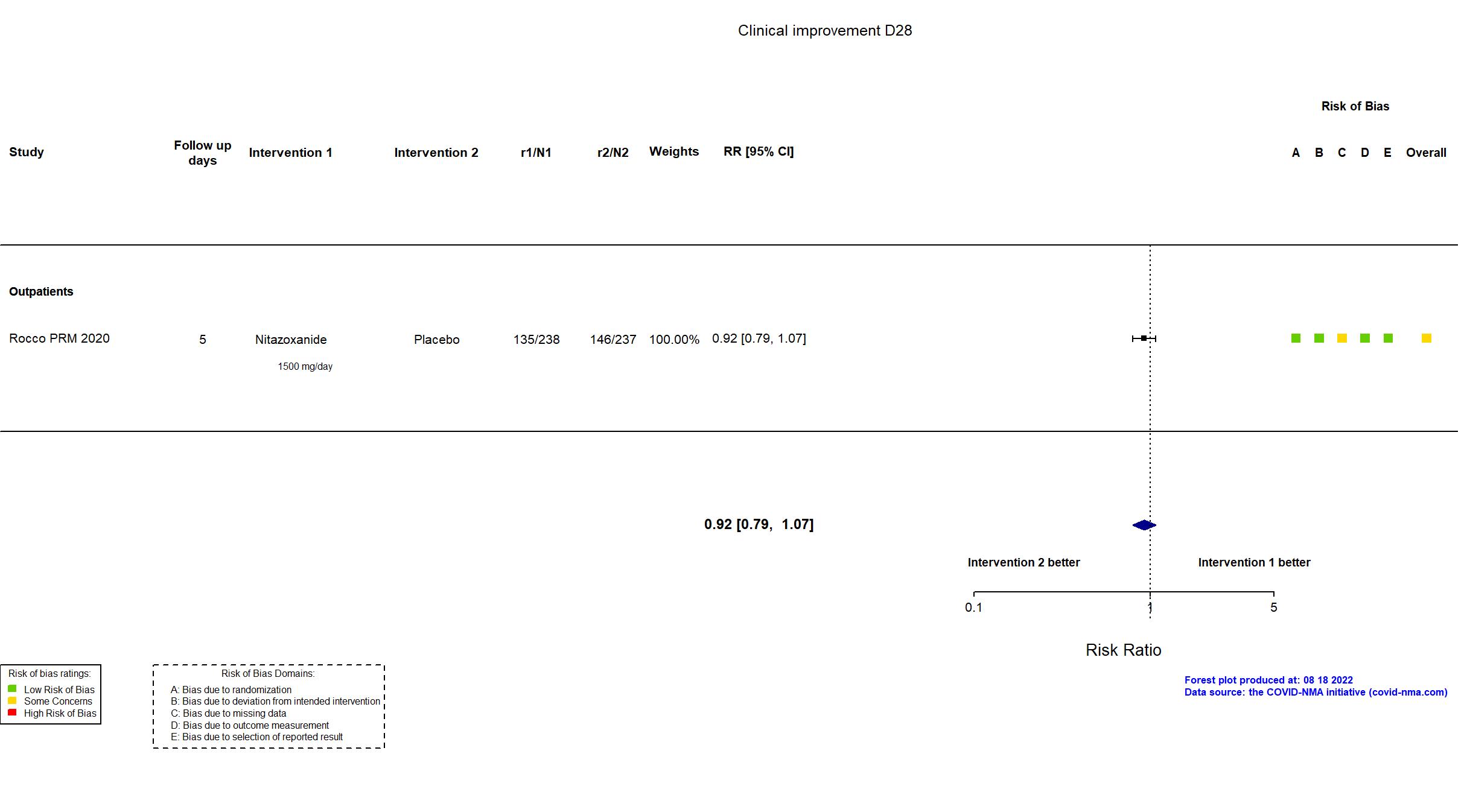











Trial NCT04498936
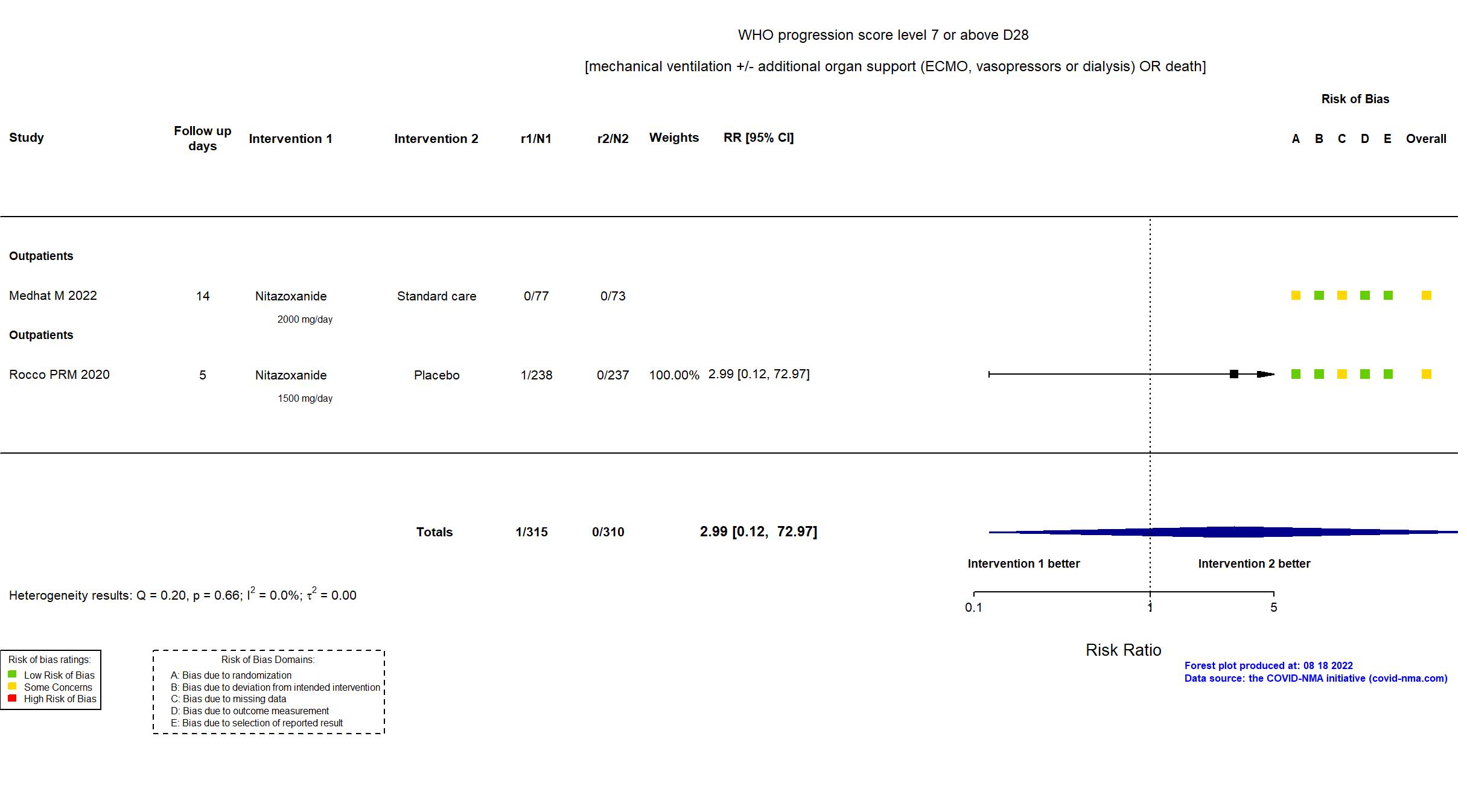
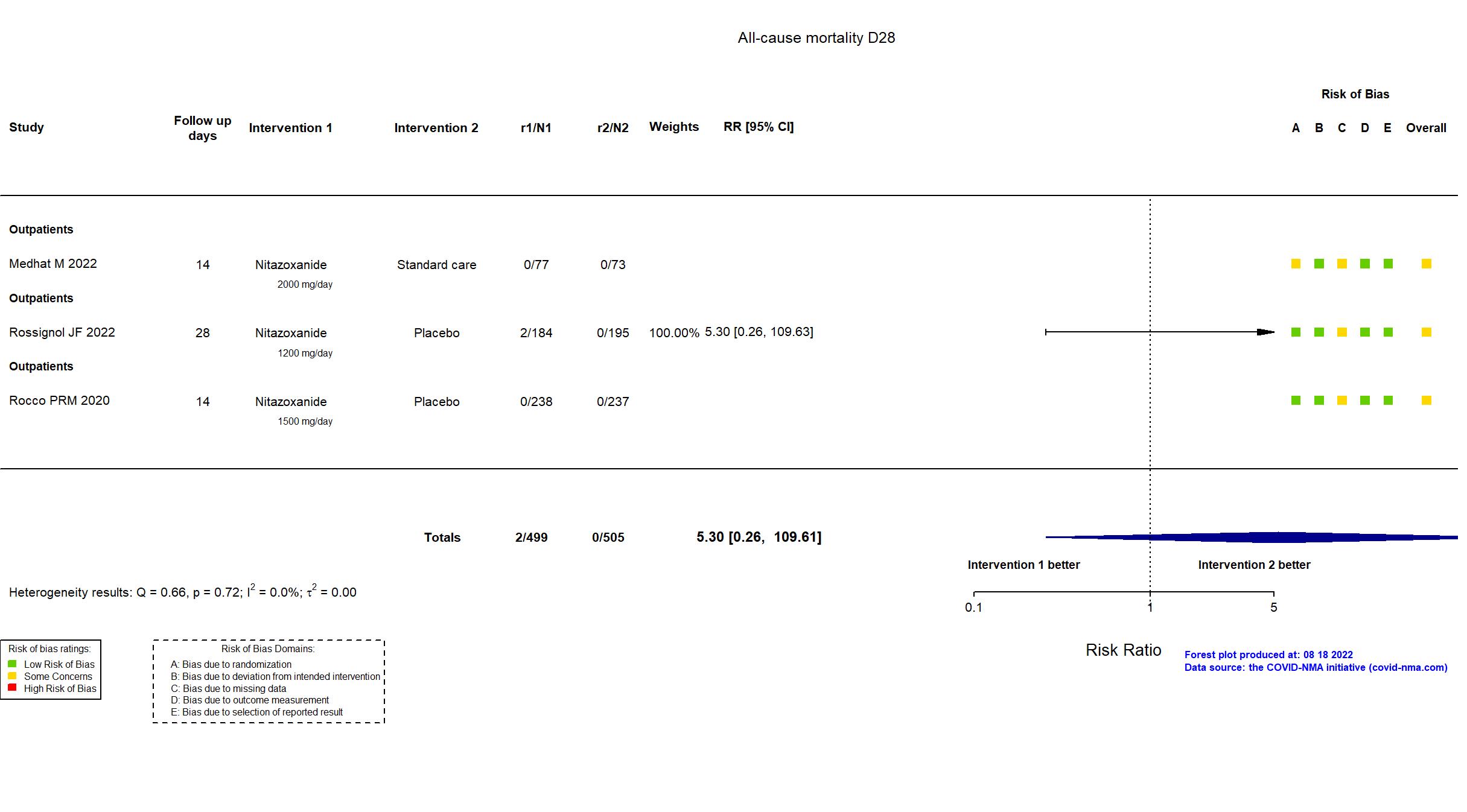
Publication Medhat M, Arab J Gastroenterol (2022) (published paper)
Dates: 2020-07-15 to 2021-10-30
Funding: Public/non profit (Egyptian Science, Technology and Innovation Funding Authority (STDF))
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / Egypt Follow-up duration (days): 14 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
SOF/LDP 400 mg + 90 mg orally once a day for 14 days Nitazoxanide 500 mg orally four times a day for 14 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : SOF/LDP=70 Nitazoxanide =77 Standard care=73 | |
| Characteristics of participants N= 220 Mean age : NR 100 males Severity : Mild: n= */ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register 1) Change of PCR from positive to negative [ Time Frame: 2 weeks ] The PCR will be done at time of recruitment, day 5, 8, 11, and 14. The time taken to have negative will be measured in each group 2) Clinical improvement [ Time Frame: 2 weeks ] Clinical improvement will be measured by detection of downgrading of cases severity according to the World Health Organization case severity classification. | |
| In the report Viral clearance | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the trial registry was used in data extraction and assessment of risk of bias. Neither protocol nor statistical analysis plan was available. Only one of the two primary outcomes in the registry is reported in the article. The registry reports an additional primary outcome, clinical improvement. The trial (n = 220) did not achieve the target sample size in the registry (n = 240). |
Trial NCT04552483
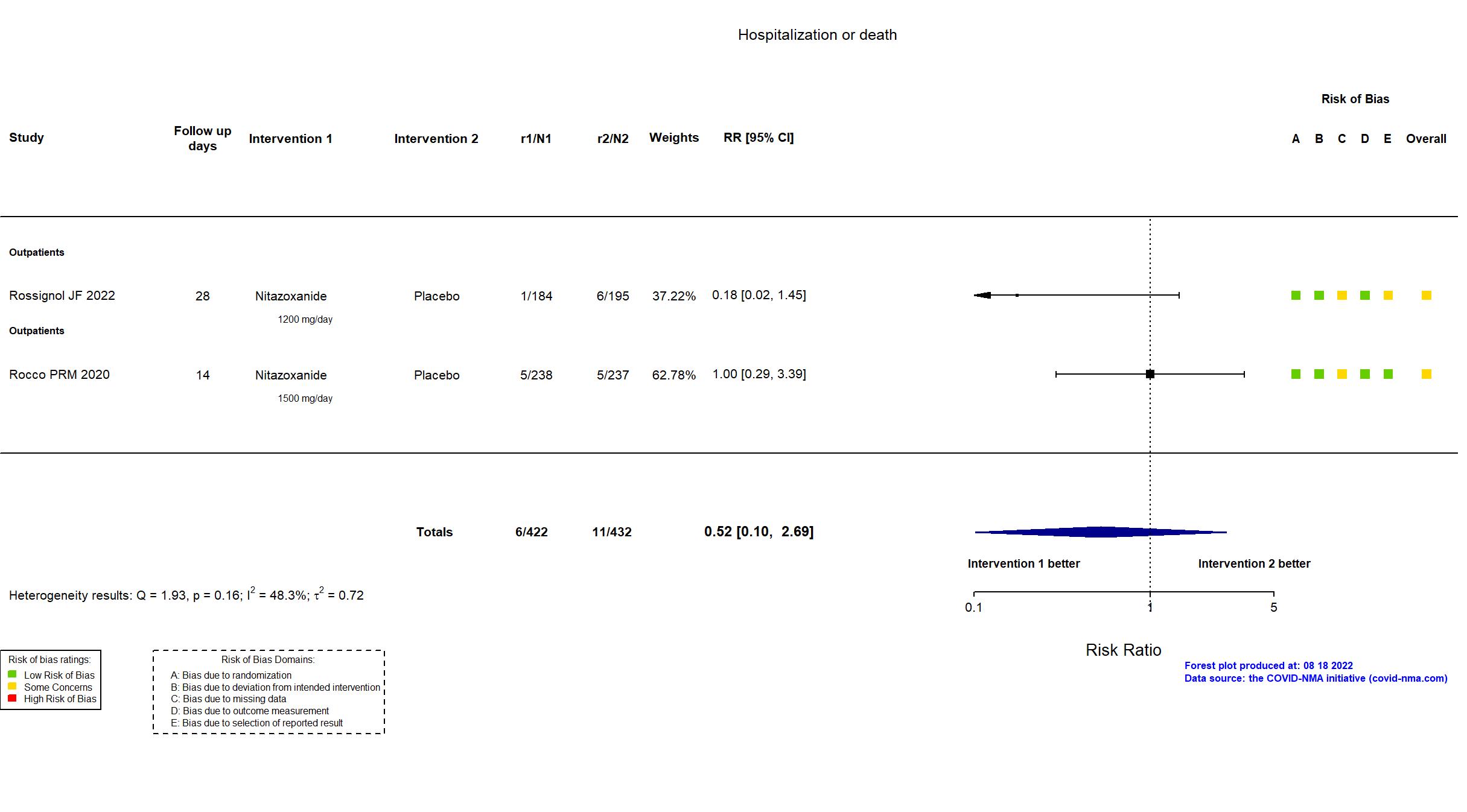
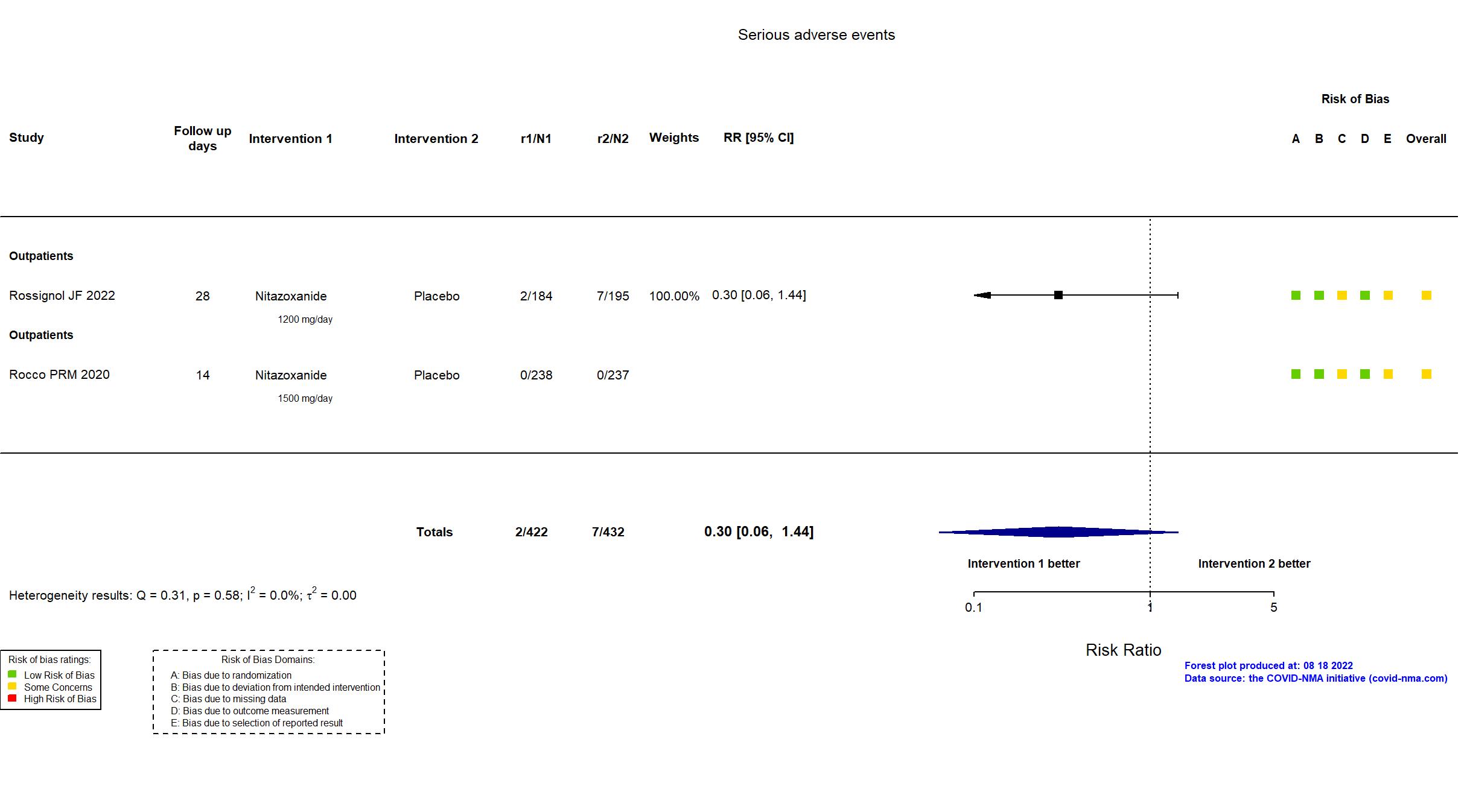
Publication Rocco PRM, Eur Respir J (2020) (published paper)
Dates: 2020-06-08 to 2020-08-20
Funding: Mixed (Brazilian Council for Scientific and Technological Development (CNPq), Brazilian Ministry of Science, Technology, and Innovation for Virus Network; Brasilia, Brazil, and Funding Authority for Studies and Projects, Brasil)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: | |
| Location :
Multicenter / Brazil Follow-up duration (days): 14 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Nitazoxanide 500 mg orally three times a day for 5 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Nitazoxanide =238 Placebo=237 | |
| Characteristics of participants N= 475 Mean age : NR 184 males Severity : Mild: n= 475/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Days with fever; Days with cough; Days with asthenia [ Time Frame: Day8 ] | |
| In the report Complete resolution of symptoms | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication:
|
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the pre-print, the study registry and supplementary materials were used in data extraction and risk of bias assessment. Study protocol and statistical analysis plan were not available at the time of data extraction. Trial registration was retrospective. Sample size justification was calculated retrospectively to describe power of the included sample size following considerable loss to follow up. The registry primary outcomes and timepoint does not reflect the combined reported primary outcome. Data for the outcome Viral negative conversion Day 7 correspond to day 5 in the trial.
This study was updated on December 3rd, 2020 after contact with authors. This study was updated on January 18th, 2021 after publication of the study report. |
Trial NCT04486313
Publication Rossignol JF, EClinicalMedicine (2022) (published paper)
Dates: 2020-08-18 to 2021-01-08
Funding: Private (Romark Institute of Medical Research)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / Puerto Rico, USA Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Nitazoxanide 600 mg orally twice a day for 5 days |
|
| Control
Placebo | |
| Participants | |
| Randomized NR Analyzed 379 participants Nitazoxanide =184 Placebo=195 | |
| Characteristics of participants N= 379 Mean age : NR 165 males Severity : Mild: n= 379/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Reducing the Time to Sustained Response [ Time Frame: Up to 21 days ] | |
| In the report Time from the first dose to sustained response (TSR), a measure of meaningful within-subject change in symptoms | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: N |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the all versions of the published and pre-print articles (SSRN and medRxiv), the prospective trial registry was used in data extraction and assessment of risk of bias. Neither study protocol nor statistical analysis plan was available. There is no change from the trial registration in the intervention and control treatments. The registry primary outcome reflects the reported primary outcome. Several outcomes reported in the article were not included in the trial registry. When all participants randomized or included for safety are considered, the study achieved its original target sample size.
Of note, participants were recruited, randomized and tested at first clinic visit with suspected COVID-19. Many were subsequently excluded from efficacy analysis for a number of reasons: a) Of 1,092 randomized, 156 subjects positive for rhinovirus/enterovirus and negative for SARS-CoV-2 at baseline were not included in ANY analysis as they are to be analyzed later as part of a separate study targeting enterovirus/ rhinovirus infection. b) 1 participant withdrew consent prior to receiving treatment, but not reported which arm. All remaining participants (n=935) who received treatment were included in the safety analysis after receiving intervention or placebo. (All outcomes extracted were in this population: mortality, AE, SAE). This study study was updated on March 9th, 2022 with data from the new versions of the preprints. This study was updated on April 27th, 2022 with data extracted from the peer-reviewed article. |