Inhaled corticosteroids vs Standard care/Placebo (RCT)
Mild outpatients
Studies included but not extracted/included in the analysis: Terada J, Drug Discov Ther, 2022
FOREST PLOTS -2022-10-07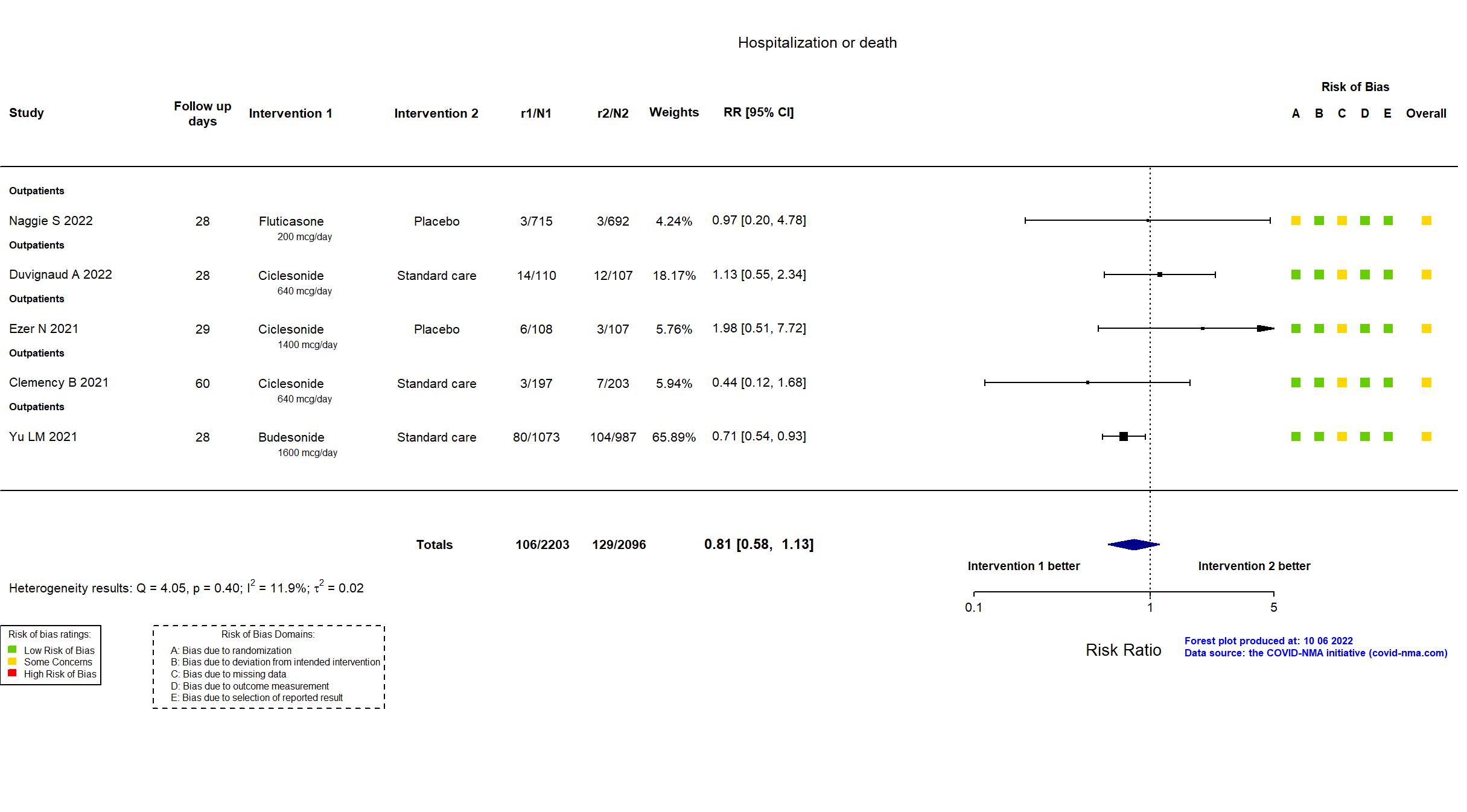
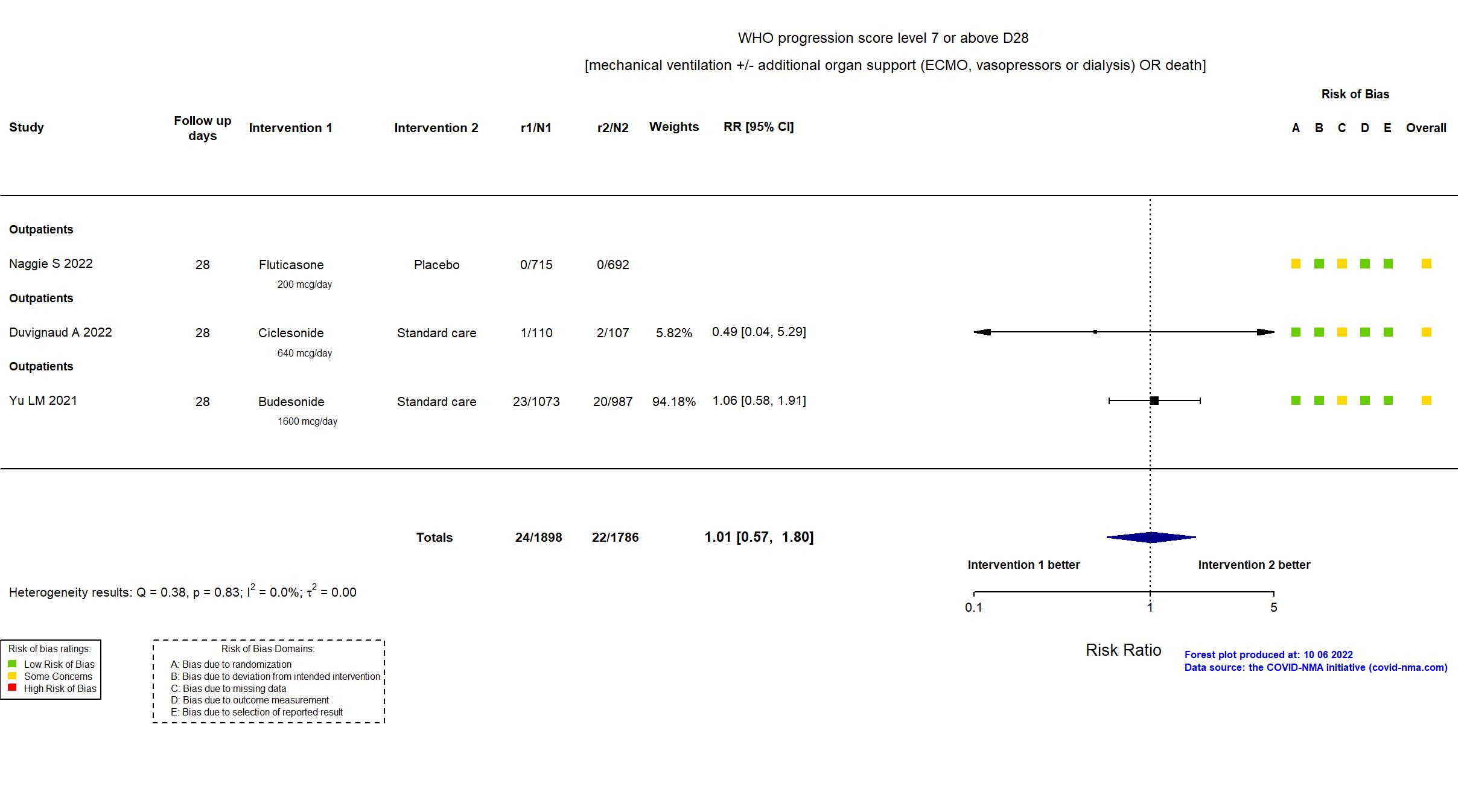
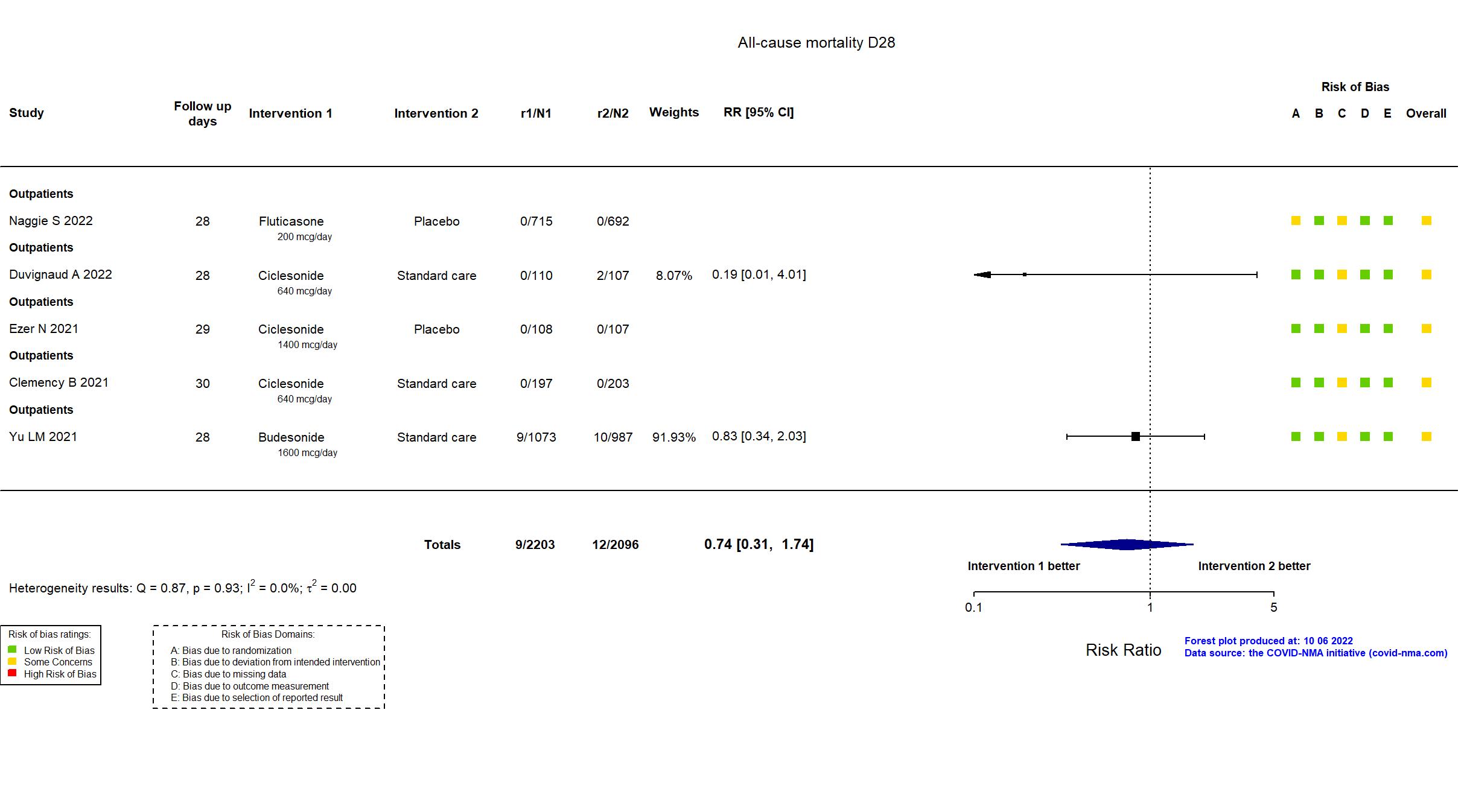
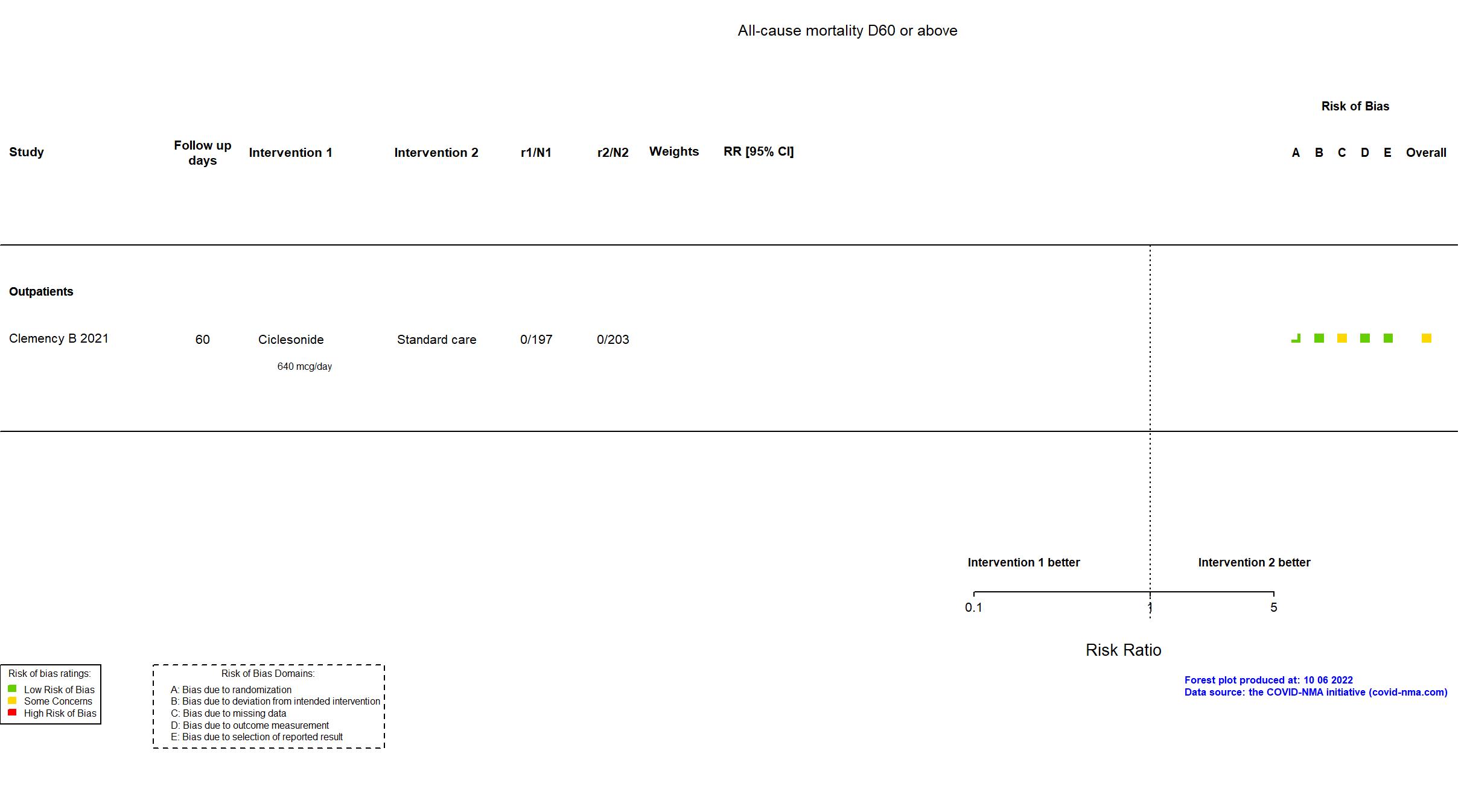
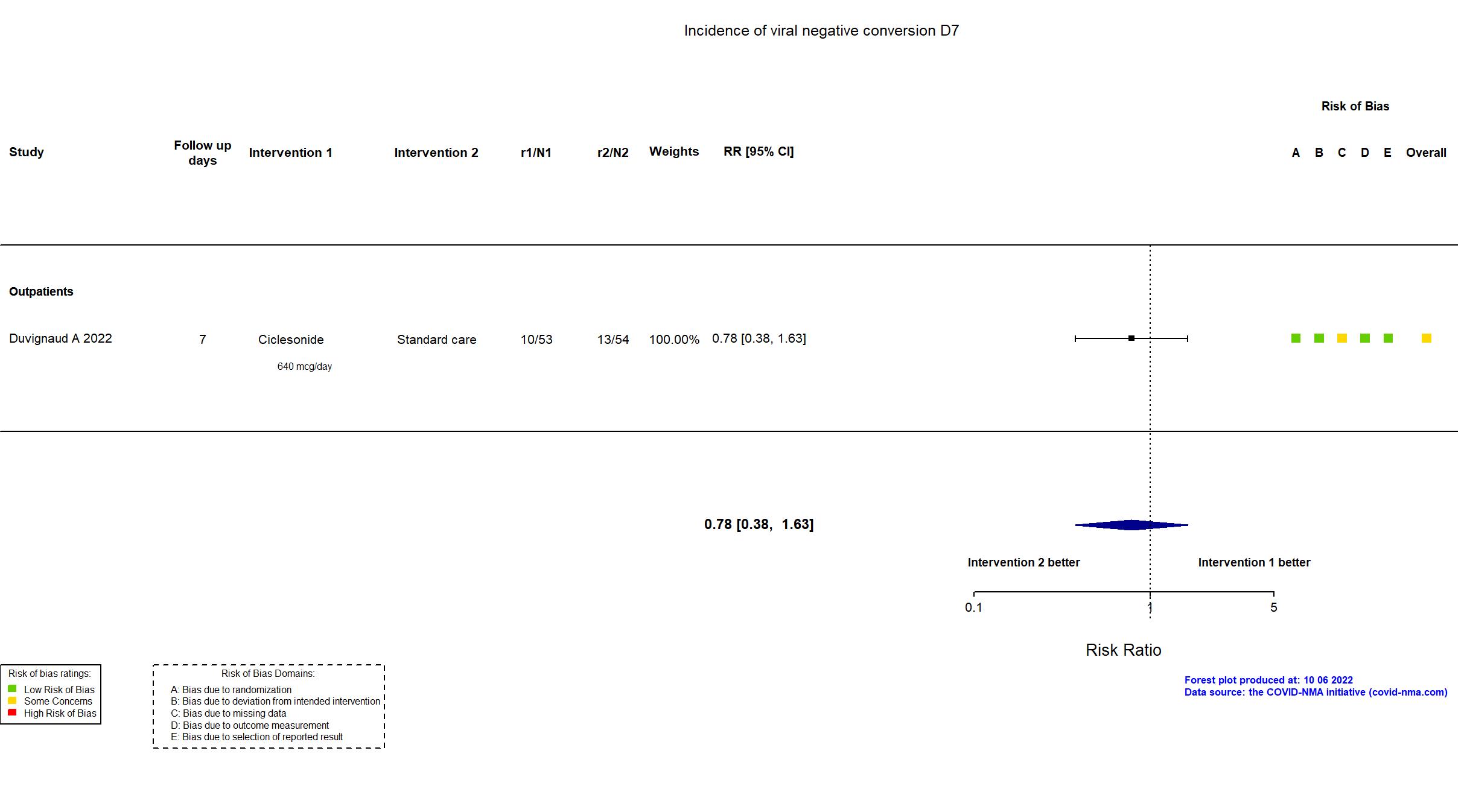
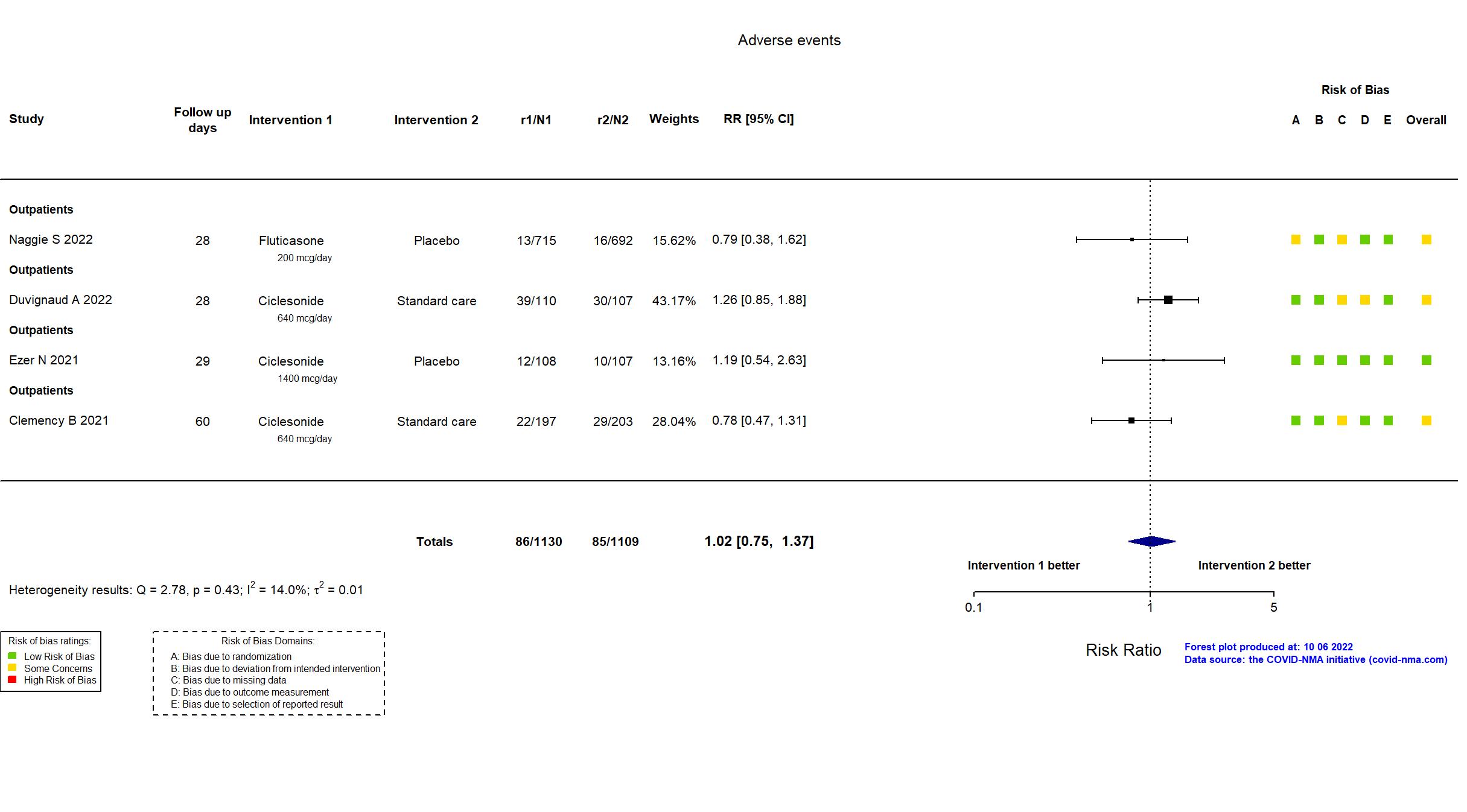
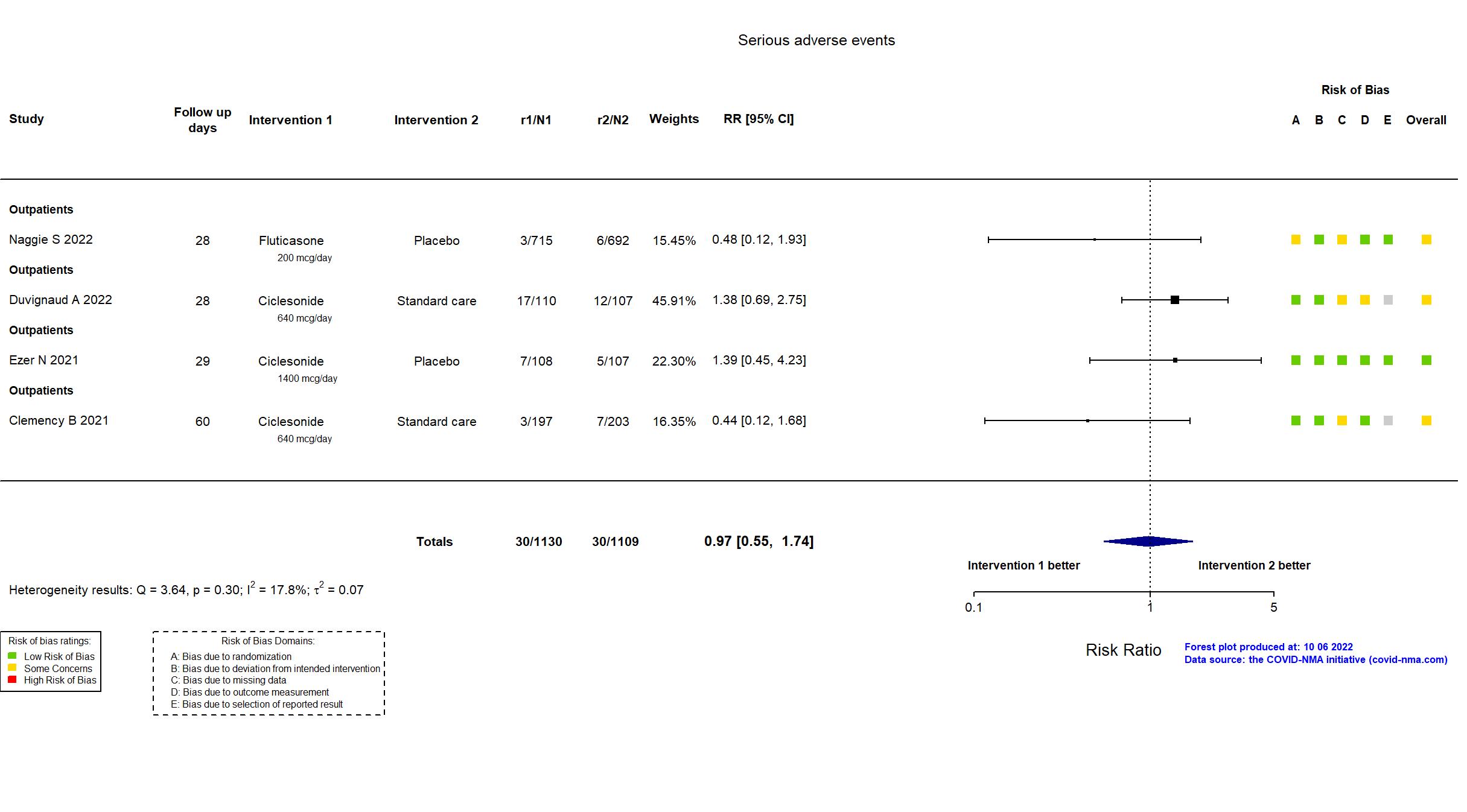

Studies included but not extracted/included in the analysis: Terada J, Drug Discov Ther, 2022
FOREST PLOTS -2022-10-07







Trial NCT04377711
Publication Clemency B, JAMA Intern Med (2021) (published paper)
Dates: 2020-06-11 to 2020-11-03
Funding: Private (Covis Pharma GmbH; National Center for Advancing Translational Sciences of the National Institutes of Health (NIH); National Heart, Lung, and Blood Institute of the NIH)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / USA Follow-up duration (days): 60 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Ciclesonide Two inhalations of 160 mcg twice a day for 30 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Ciclesonide=197 Standard care=203 | |
| Characteristics of participants N= 400 Mean age : NR 179 males Severity : Mild: n= 400/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Time to alleviation of COVID-19-related symptoms by Day 30 [ Time Frame: Day 30 ] | |
| In the report Time to alleviation of all COVID-19–related symptoms (cough, dyspnea, chills, feeling feverish, repeated shaking with chills, muscle pain, headache, sore throat, and new loss of taste or smell) by day 30 | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the published article, the pre-print, the trial registry, protocol and statistical analysis plan were used in data extraction and assessment of risk of bias. The primary outcome in the article reflects that in the registry/protocol. The primary outcome was changed prior to start of recruitment from emergency department visits or hospital admission to symptom resolution. The study achieved its target sample size.
This study was updated on the 15th of December, 2021 with data from the published article. This study was updated on September 28th, 2022 with new data extracted from the trial registry. |
Trial NCT04356495, EudraCT 2020-001435-27
Publication COVERAGE - Duvignaud A, Clin Microbiol Infect (2022) (published paper)
Dates: 2020-12-29 to 2021-07-23
Funding: Public/non profit (French Ministry of Health, French National Research Agency, University of Bordeaux, Inserm/REACTing)
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / France Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Ciclesonide 2 puffs (160 mcg each) by inhalation twice a day for 10 days |
|
| Control
Standard care vitamin supplementation (AZINC vitality®, 2 pills per day) for 10 days | |
| Participants | |
| Randomized participants : Ciclesonide=110 Standard care=107 | |
| Characteristics of participants N= 217 Mean age : NR 106 males Severity : Mild: n= 217/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register 1) Proportion of participants who had a Grade 3 or 4 adverse event [ Time Frame: From inclusion (day0) to day 14 ]; 2) Efficacy phase: Death [ Time Frame: From inclusion (day0) to day 14 ], Efficacy phase: oxygen therapy [ Time Frame: From inclusion (day0) to day 14 ], Efficacy phase: hospitalization [ Time Frame: From inclusion (day0) to day 14 ] | |
| In the report 1) occurrence of grade 3-4-5 adverse events; 2) combination of hospitalisation, need for COVID19-related oxygen therapy at home or death | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the published article, the protocol, statistical analysis plan, supplementary appendices and study registry were used in data extraction and risk of bias assessment. There is no change from the trial registration in the intervention and control treatments. The registry primary outcome reflects the reported primary outcome. The study (n = 217) did not achieve its target sample size (n = 702) because recruitment was terminated after futility was determined in a pre-planned interim analysis.
This study was updated on September 1st, 2022 with data extracted after contact with authors. |
Trial NCT04435795
Publication CONTAIN - Ezer N, BMJ (2021) (published paper)
Dates: 2020-09-15 to 2021-06-08
Funding: Mixed (McGill University Health Centre Foundation and the McGill Interdisciplinary Initiative in Infection and Immunity. The placebo metered dose inhaler was donated by GlaxoSmithKline. Nasal saline, and spacers for the inhalers were purchased by NE. The study drug was donated by COVIS Pharma. )
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / Canada Follow-up duration (days): 29 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Ciclesonide 600 mcg inhaled twice a day + 200 mcg intranasally once a day for 14 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Ciclesonide=108 Placebo=107 | |
| Characteristics of participants N= 215 Mean age : NR 94 males Severity : Mild: n= 203/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Proportion of participants with no symptoms of cough, fever or dyspnea [ Time Frame: day 7 ] | |
| In the report Resolution of self-reported fever and all respiratory symptoms at day 7 of treatment. Respiratory symptoms included cough (wet or dry) or dyspnoea (which included the description of shortness of breath, chest congestion, or chest tightness as synonyms). | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the published article, the protocol, the statistical analysis plan, the supplementary and the study registry were used in data extraction and risk of bias assessment. The study (n=215) did not achieve the target sample size (n=318) and was terminated early for expected futility to meet total enrolment. There is no change from the trial registration in the intervention and control treatments. The primary outcome indicated in registry reflects the primary outcome reported in the paper. Adverse events were reported in the paper but were not specified in the registry.
This study was updated on March 16th, 2022 with data extracted after contact with authors. |
Trial NCT04885530
Publication ACTIV 6 - Naggie S, medRxiv (2022) (preprint)
Dates: 2021-08-10 to 2022-02-12
Funding: Mixed (National Center for Advancing Translational Sciences (NCATS); Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority; GSK provided the product used in the study.)
Conflict of interest: No
| Methods | |
| RCT Blinding: triple blinding | |
| Location :
Multicenter / USA Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Fluticasone 200 mcg by inhalation once a day for 14 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Fluticasone=715 Placebo=692 | |
| Characteristics of participants N= 1407 Mean age : NR 470 males Severity : Mild: n= 1277/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Number of hospitalizations as measured by patient reports. [ Time Frame: Up to 28 days ]; Number of deaths as measured by patient reports [ Time Frame: Up to 28 days ]; Number of symptoms as measured by patient reports [ Time Frame: Up to 28 days ] | |
| In the report Time to recovery, defined as the third of 3 consecutive days without symptoms. | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the pre-print article, the trial registry and supplementary appendices were used in data extraction and assessment of risk of bias. Neither protocol no statistical analysis plan was available. There is no change from the trial registration in the intervention and control treatments. The primary outcome in the article (time to recovery) differed from the three primary outcomes available in the platform study in the in the registry (number of hospitalizations, deaths and symptoms). A substantial proportion (9.2%) of those randomized were not included in follow up and analysis because they did not receive the study interventions due to the study’s decentralized design and reliance on home delivery. Nevertheless, the trial (n = 1277) achieved its target sample size (n = 1200). |
Trial NCT04416399
Publication STOIC - Ramakrishnan S, Lancet Respir Med (2021) (published paper)
Dates: 2020-07-16 to 2020-12-09
Funding: Mixed (Oxford NIHR Biomedical Research Centre; AstraZeneca )
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Single center / UK Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Budesonide Two 400 mcg inhalations twice a day for up to 28 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Budesonide=73 Standard care =73 | |
| Characteristics of participants N= 146 Mean age : NR 62 males Severity : Mild: n= 146/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register Emergency department attendance of hospitalisation related to COVID-19 [ Time Frame: Day 1 to day 28 ] | |
| In the report COVID-19-related Urgent Care visit, Emergency Department assessment or hospitalisation | |
| Documents avalaible |
Protocol NR Statistical plan Yes Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
* |
| General comment |
No review-specific outcome data extracted. Pending contact with authors.
In addition to the published/pre-print article, the trial registry, statistical analysis plan and supplementary appendix were used in data extraction and assessment of risk of bias. The study protocol was not available. The trial was stopped early after independent statistical review concluded that study outcome would not change with further participant enrolment. As a result, the target sample size specified in the registry was not achieved. There were no differences between the trial registration and the article in the intervention and control treatments or the participant inclusion and exclusion criteria. Outcomes listed in the registry were reported, but none were relevant for the COVID-19 NMA. Consequently, no outcome data have been extracted for this study. On 13th of April ,2021, this study was updated based on the published report. |
Trial ISRCTN86534580; EudraCT 2020-001209-22
Publication PRINCIPLE - Yu LM, Lancet (2021) (published paper)
Dates: 2020-11-27 to 2021-03-31
Funding: Public/non profit (UK Research and Innovation and the Department of Health and Social Care through the National Institute for Health Research)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / UK Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Budesonide 800 mcg (2 x 400 mcg inhalations) twice a day for 14 days. |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Budesonide =1073 Standard care=NR | |
| Characteristics of participants N= 1073 Mean age : NR 835 males Severity : Mild: n= 1719/ Asymptomatic: n=0 | |
| Primary outcome | |
| In the register 1. Time taken to self-reported recovery, defined as the first instance that a participant reports feeling recovered from possible COVID-19; 2. Hospitalisation and/or death. Both collected within 28 days of randomization (ISRCTN). | |
| In the report 1) Time to first reported recovery defined as the first instance that a participant reports feeling recovered; and 2) Hospitalization or death related to COVID-19. | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication:
|
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the published/pre-print article, the trial registry, protocol and statistical analysis plan were used in data extraction and assessment of risk of bias. PRINCIPLE is an adaptive platform trial. The primary outcome was changed from hospitalization or death within 28 days to first instance that a participant reports feeling recovered on the recommendation of the Trial Management Group and Trial Steering and with ethical approval due to low incidence of the original outcome. The Data Monitoring and Safety Committee determined that the pre-specified superiority criterion was met on time to recovery, in both the SARS-CoV-2 positive population and the overall study population. Recruitment was stopped because the Trial Steering Committee decided that accumulating further data to reach pre-specified futility or superiority criteria on hospitalization/death was unlikely due to the successful vaccine rollout and lower than originally anticipated event rate. Therefore the study did not achieve the target sample size specified in the trial registry.
On 16th of August, 2021, this study was updated based on the published report. |