Triazavirin vs Placebo (RCT)
Hospitalized patients
FOREST PLOTS -2021-06-18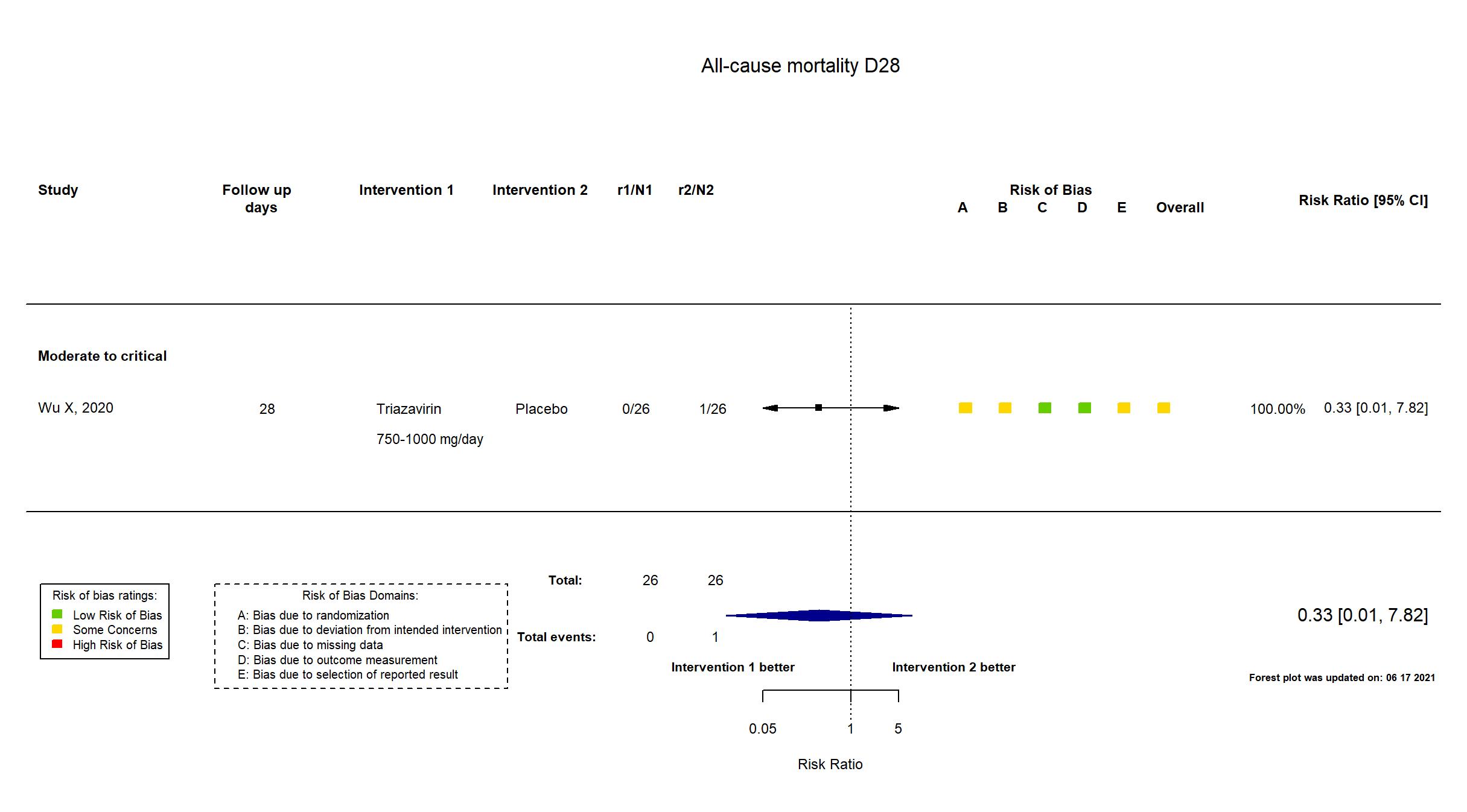
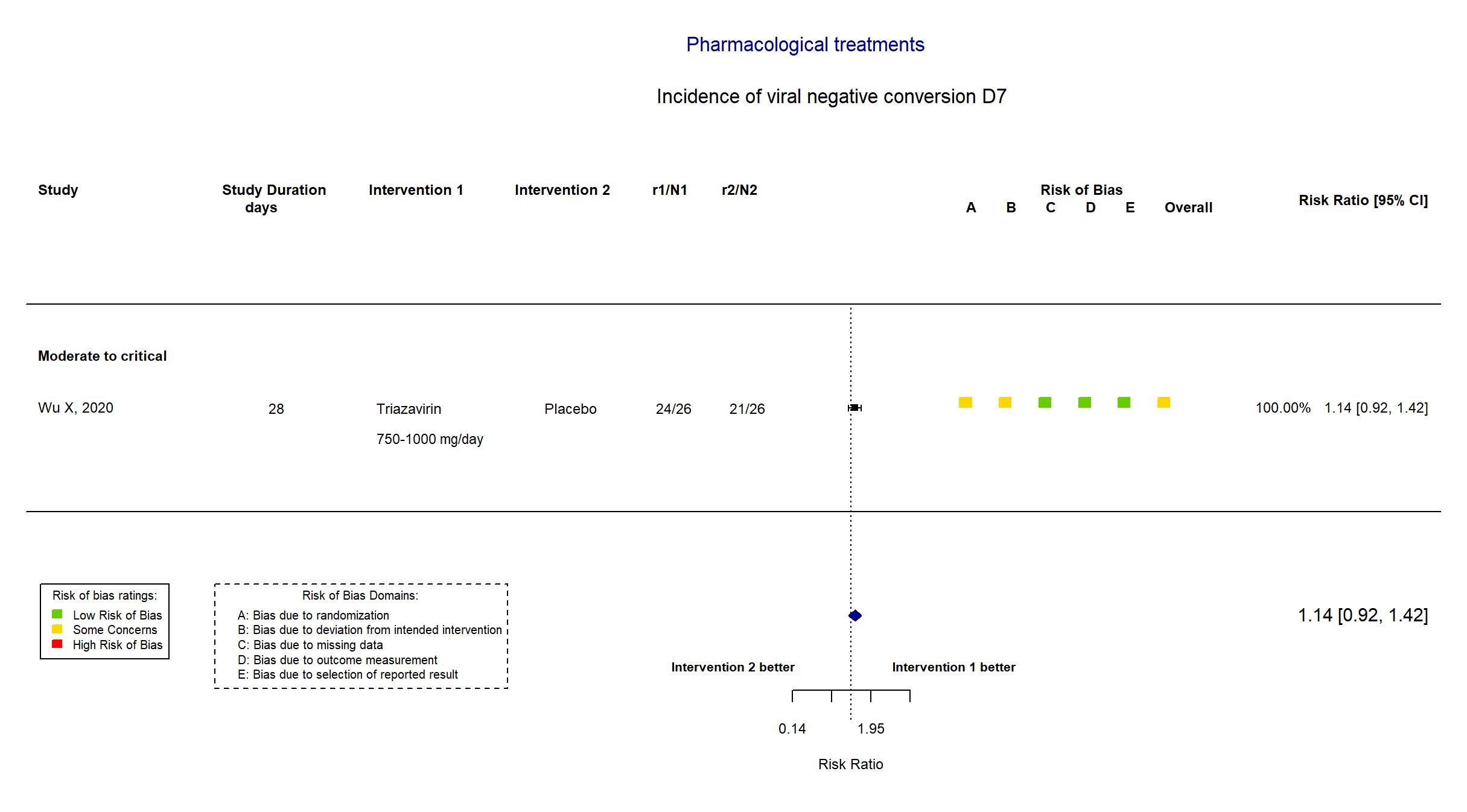
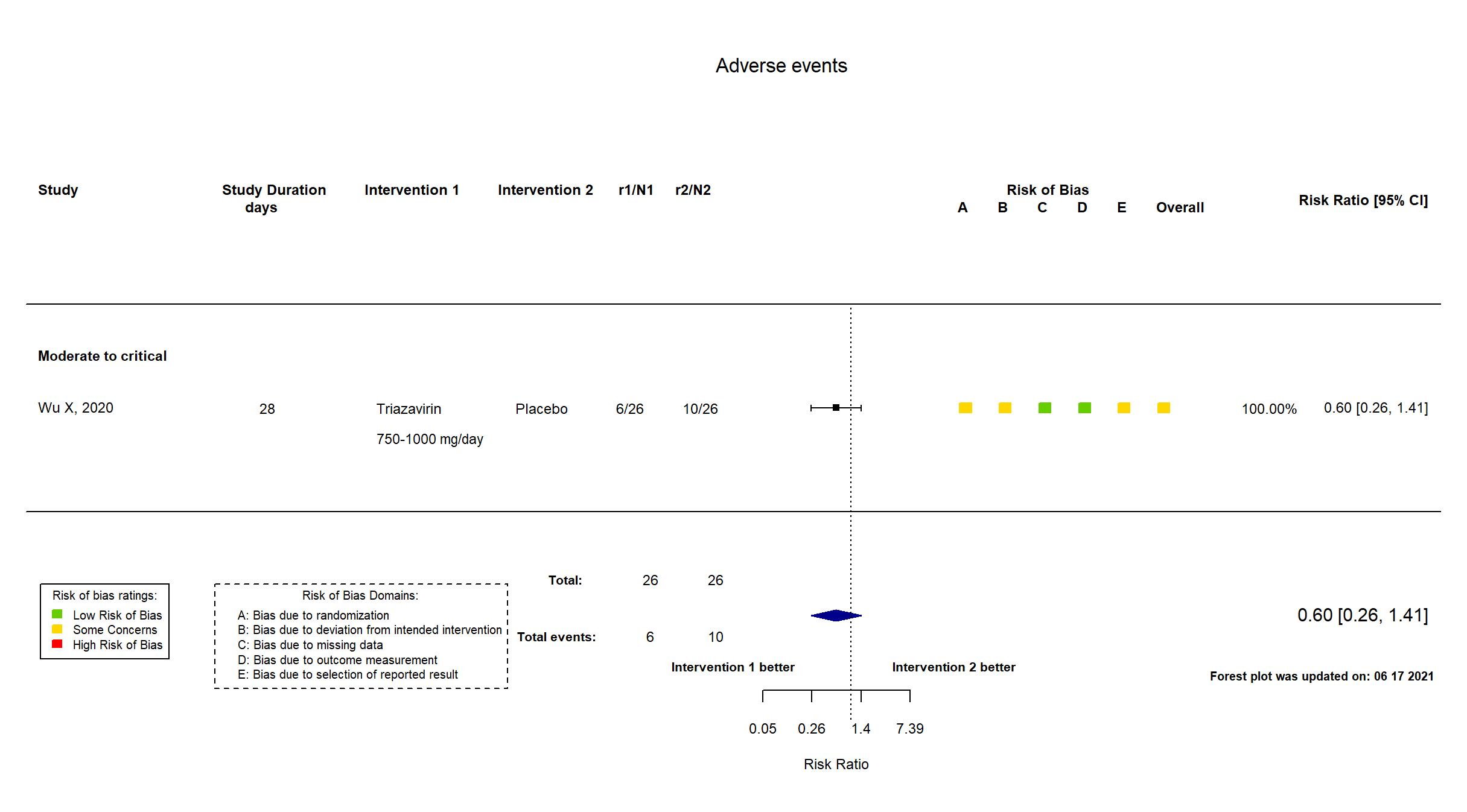
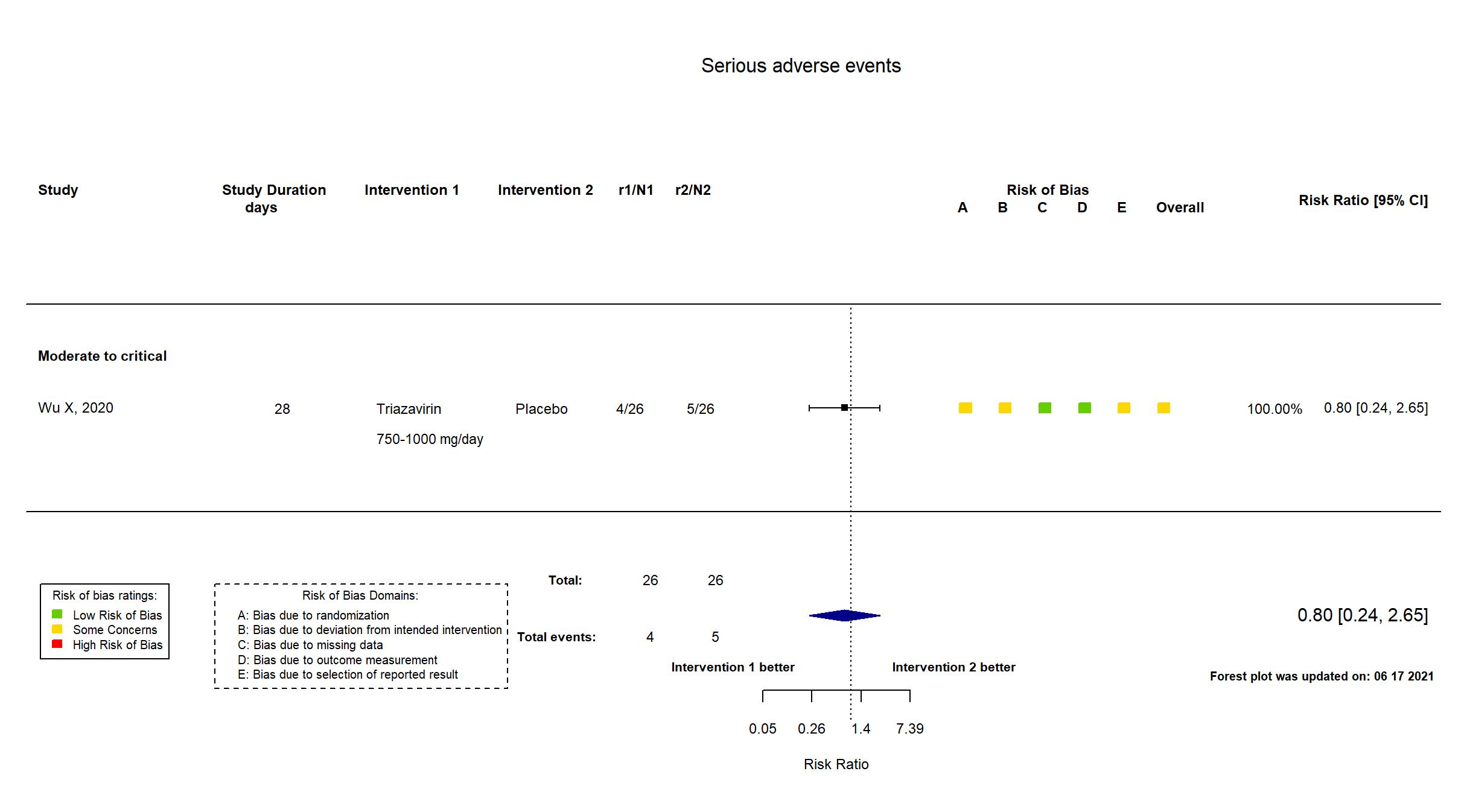




Trial ChiCTR2000030001
Publication Wu X, Engineering (2020) (published paper)
Dates: 2020-02-14 to 2020-03-06
Funding: Public/non profit (Chinese Academy of Engineering Projects for COVID-19; Heilongjiang Province Urgent Project-6 for COVID-19)
Conflict of interest: No
| Methods | |
| RCT Blinding: Participants and outcome assessor | |
| Location :
Multicenter / China Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Triazavirin 250 mg orally 3 times a day (or 4 times if severe or critical patient) for 7 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Triazavirin=26 Placebo=26 | |
| Characteristics of participants N= 52 Mean age : NR 26 males Severity : Mild: n=* / Moderate: n=* / Severe: n=* Critical: n=* | |
| Primary outcome | |
| In the register Time to Clinical recovery | |
| In the report Time to clinical improvement | |
| Documents avalaible |
Protocol Yes. In English Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the protocol and the study registry were used for data extraction and risk of bias assessment (neither registered before initiation of study). The trial was terminated ahead of schedule by the decision of the data and safety monitoring board because the COVID-19 outbreak in China was under control at the time, and there had been no new cases for one week. As a result, the target sample size specified in the registry/protocol was not achieved (planned sample size was 240). There is no change from the trial registration in the intervention and control treatments. The primary outcome indicated in registry/protocol reflects the primary outcome reported in the paper. |