Hyperimmune anti-COVID-19 Intravenous Immunoglobulin vs Standard care/Placebo (RCT)
Hospitalized patients
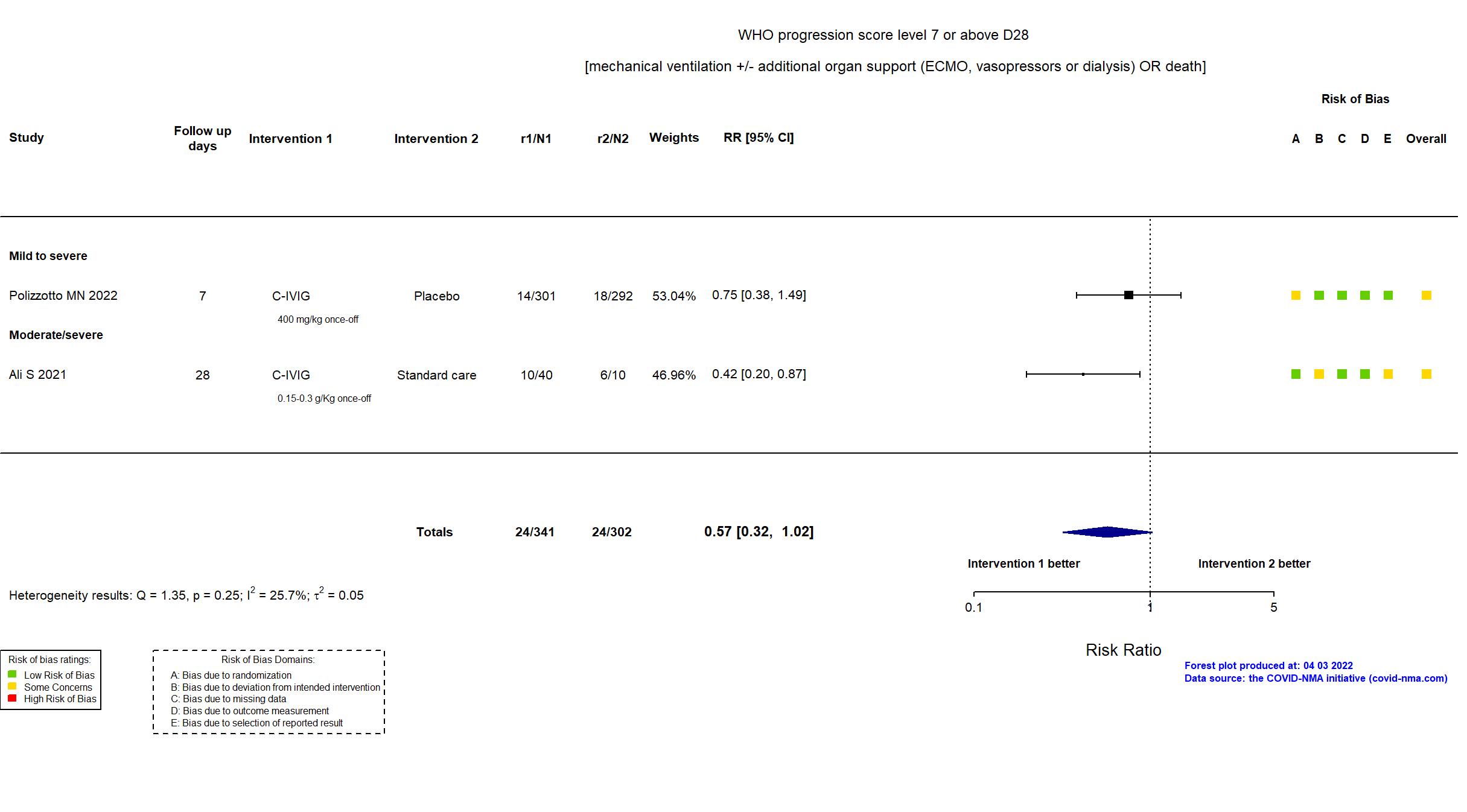
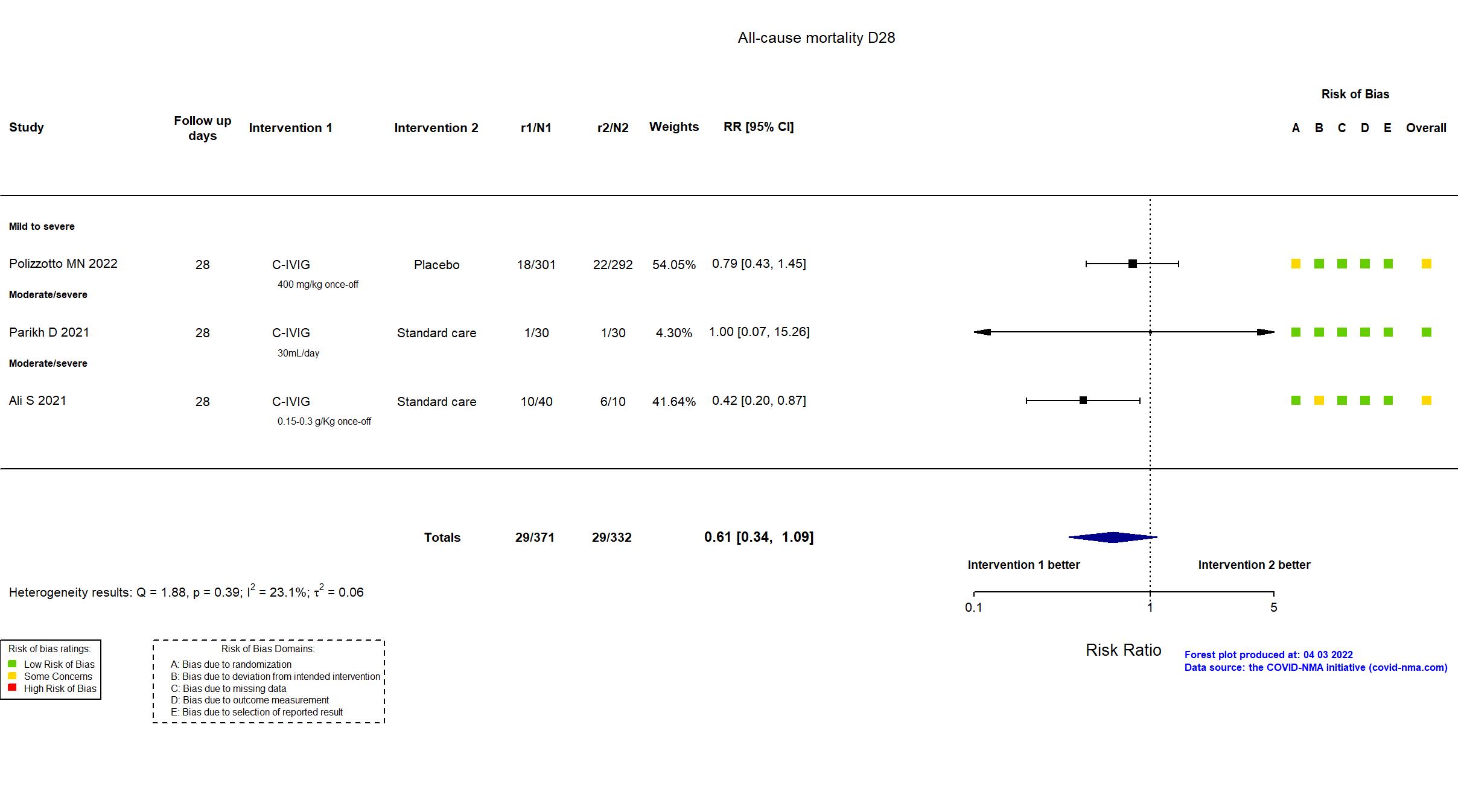
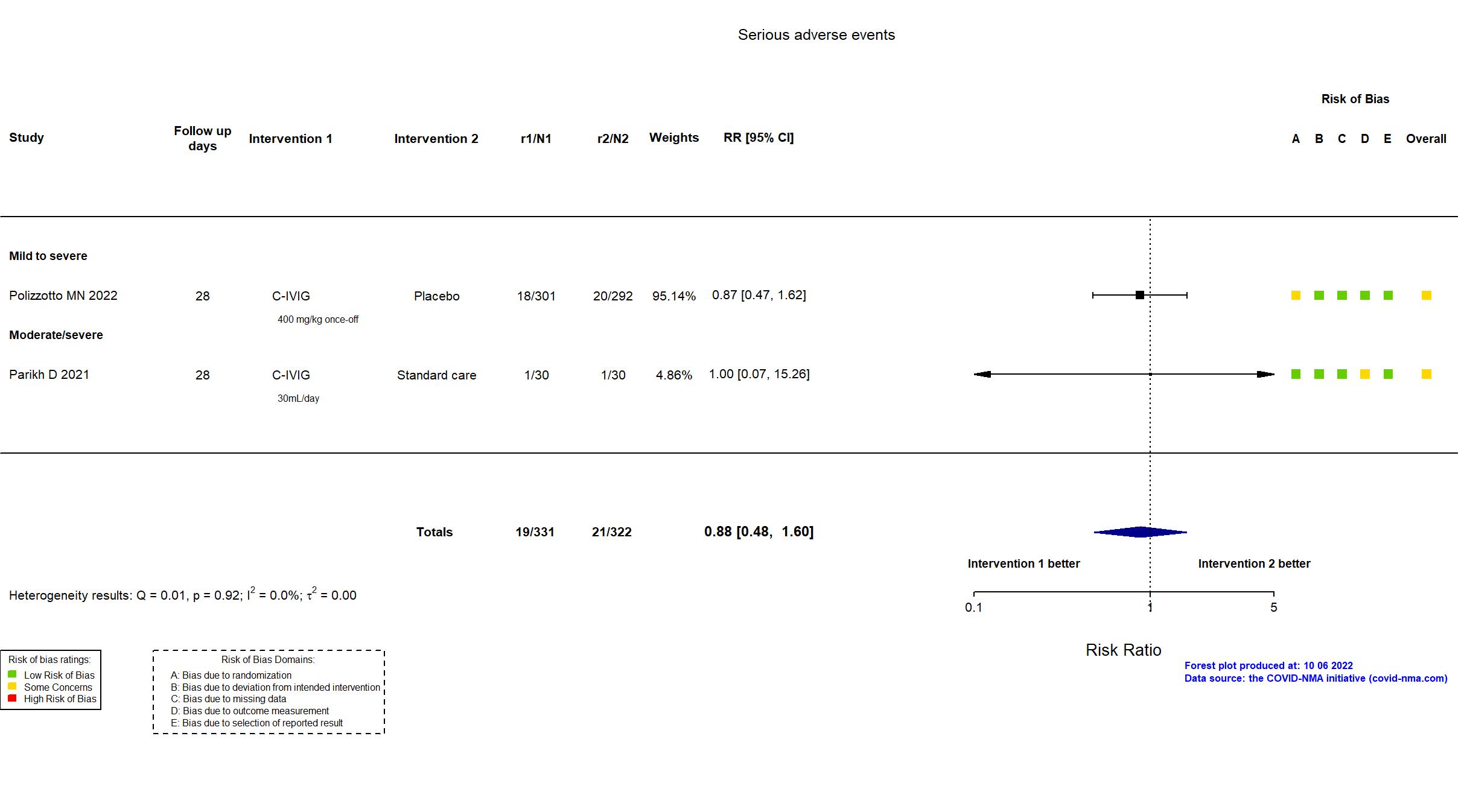
FOREST PLOTS -2022-10-07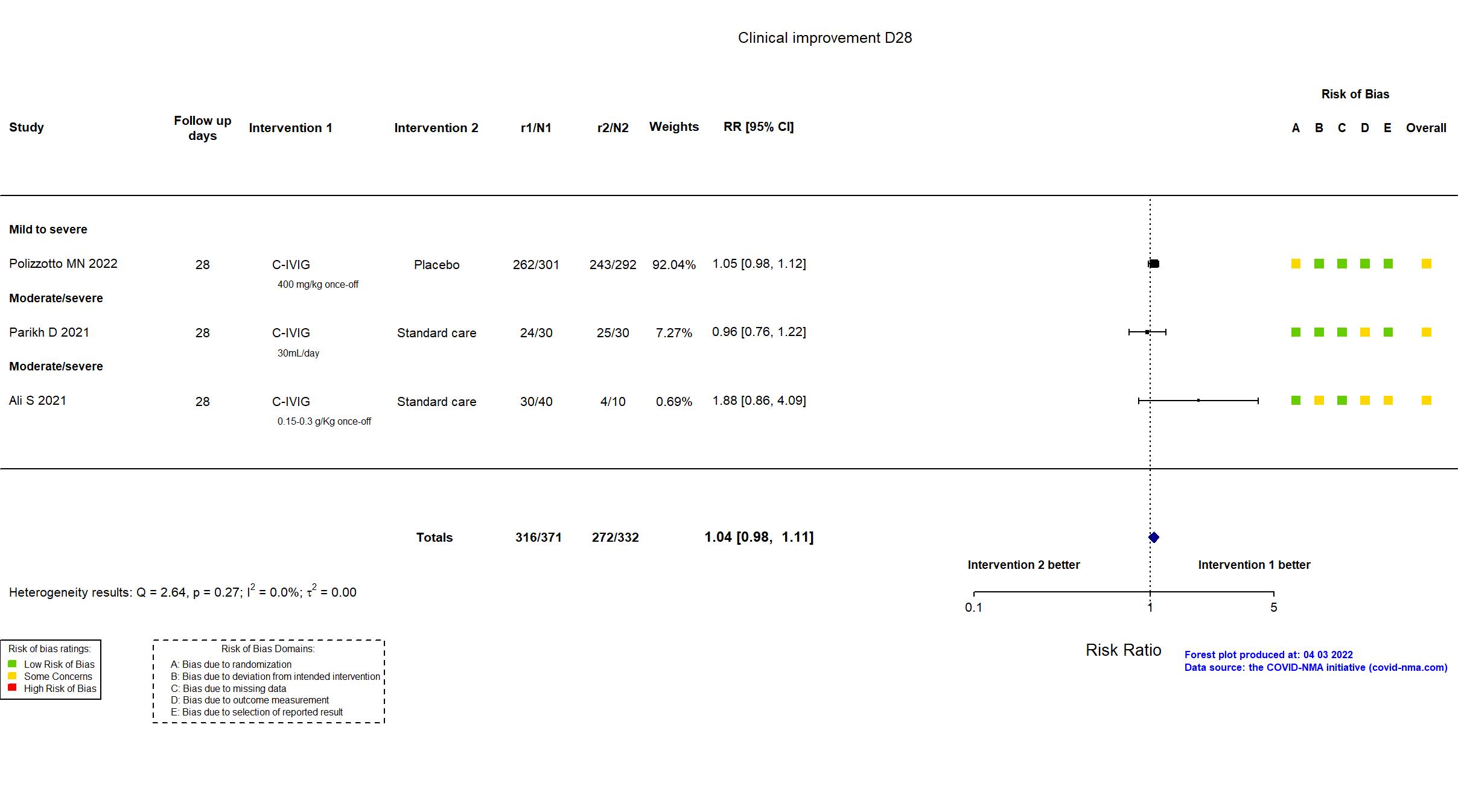









Trial NCT04521309
Publication Ali S, EClinicalMedicine (2021) (published paper)
Dates: 2020-06-19 to 2021-02-03
Funding: Public/non profit (Higher Education Commission (HEC), Pakistan)
Conflict of interest: No
| Methods | |
| RCT Blinding: single blinding | |
| Location :
Single center / Pakistan Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
C-IVIG 0.15-0.3 g/Kg IV once-off |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : C-IVIG=40 Standard care=10 | |
| Characteristics of participants N= 50 Mean age : NR 35 males Severity : Mild: n=0 / Moderate: n=22 / Severe: n=27 Critical: n=1 | |
| Primary outcome | |
| In the register 28 Days mortality; Requirement of supplemental oxygen support; Number of days on assisted ventilation; Days to step down; Days to Hospital Discharge; Adverse events during hospital stay; Change in C-Reactive Protein (CRP) levels; Change in neutrophil lymphocyte ratio | |
| In the report 28-day mortality; clinical status; PaO2/FiO2 ratio | |
| Documents avalaible |
Protocol Yes. In English Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the study registry and protocol were used in data extraction and risk of bias assessment. The registration was made 2 months after start of recruitment, but nearly 6 months before the end of the study, with no changes to outcomes. The dosages in the registry were changed after completion of the study. The primary outcomes in the report (28-day mortality, patient's clinical status during study duration and Horowitz index at outcome day) are not the same as those in the registry (28 days mortality, requirement of supplemental oxygen support, number of days on assisted ventilation, days to step down and to hospital discharge, adverse events during hospital stay, change in C-Reactive Protein levels, and change in neutrophil lymphocyte ratio; time frame: 28 days). The ordinal scale on which discharge and WHO score 7 or above outcome data are based was not included in the registry or protocol. The article reports a small phase I/II trial that achieved its target sample size. |
Trial CTRI/2020/09/027903
Publication Parikh D, Indian Pract (2021) (published paper)
Funding: Private (Intas Pharmaceuticals Ltd., India)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / India Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
C-IVIG 30 mL IV on day 1 and 2 |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : C-IVIG=30 Standard care=30 | |
| Characteristics of participants N= 60 Mean age : NR 44 males Severity : Mild: n=0 / Moderate: n=37 / Severe: n=23 Critical: n=0 | |
| Primary outcome | |
| In the register Mean change from Day 1 to Day 8 in clinical outcome of treatment with COVID-19 Hyper-Immuneglobulin (Human) as compared to the control arm as assessed by 8-point ordinal scale | |
| In the report mean change from day 1 to day 8 in an 8-point ordinal scale | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the preprint and published articles, the registry (dated Sept 18, 2020) was used for data extraction and analysis. The study achieved the target sample size specified in the trial registry. There is no change from the trial registration in the intervention and control treatments. The primary outcome indicated in registry reflects the primary outcome reported in the paper however, the preprint also reports on incidence of viral conversion (D7) which was not prespecified.
The study was updated on March 17th, 2022 with data extracted from contact with authors. The study was also published in the Indian Practitioner Journal. Data from this is presented. |
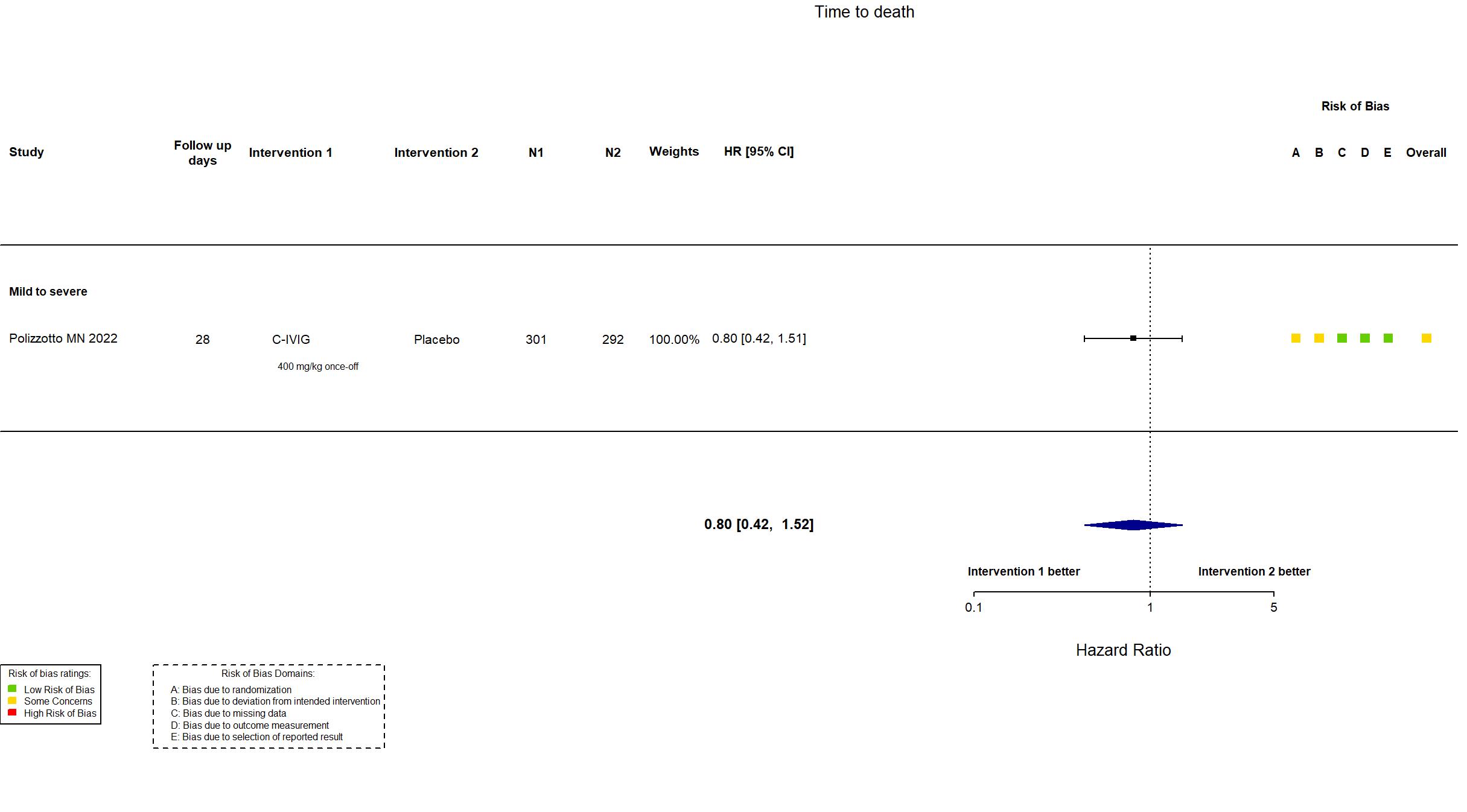
Trial NCT04546581
Publication ITAC (INSIGHT 013) - Polizzotto MN, Lancet (2022) (published paper)
Dates: 2020-10-08 to 2021-02-10
Funding: Mixed (US National Institutes of Health. Trial medications were donated by CSL Behring, Emergent BioSolutions, Grifols, Takeda, and Gilead Sciences. Many authors were employees of these companies.)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: triple blinding | |
| Location :
Multicenter / Argentina, Denmark, Germany, Greece, Indonesia, Israel, Japan, Nigeria, Spain, UK, USA. Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
C-IVIG 400 mg/kg once-off, capped at 40 g. Commenced at rate of 0·5 mg/kg per min for 30 mins; if tolerated, doubled at 30 min intervals to maximum of 4 mg/kg per min. |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Placebo=292 C-IVIG=301 | |
| Characteristics of participants N= 593 Mean age : NR 336 males Severity : Mild: n=166 / Moderate: n=369 / Severe: n=58 Critical: n=0 | |
| Primary outcome | |
| In the register Ordinal Outcome Scale - Day 7 [ Time Frame: 7 days ] The primary objective is to compare the clinical status of patients in each group on day 7 of follow-up using the primary ordinal outcome with 7 mutually exclusive categories: 7. Death 6. End-organ failure 5. Life-threatening end-organ dysfunction 4. Serious end-organ dysfunction 3. Moderate end-organ dysfunction 2. Limiting symptoms due to COVID-19 1. No limiting symptoms due to COVID-19. Outcome is reported as the percent of participants in each of 7 categories. | |
| In the report An ordinal outcome based on the patient's clinical status on day 7. The seven categories of this outcome ranged from return to usual activities with no more than minimal symptoms due to COVID-19, to death. Primary safety outcome was a composite of death, serious adverse events, and grade 3 or 4 adverse events up to day 7. | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the published article, the registry, protocol, statistical analysis plan and supplementary appendices were used in data extraction and assessment of risk of bias. The primary outcome in the article reflects that in the registry. The study (n = 593) achieved its target sample size (n = 500).
This study was updated on September 28th, 2022 with data extracted from the registry (SAE results). |