Ruxolitinib vs Placebo (RCT)
Hospitalized patients
Studies included but not extracted/included in the analysis: Rein L, Crit Care Med, 2022
FOREST PLOTS -2022-05-16


Studies included but not extracted/included in the analysis: Rein L, Crit Care Med, 2022
FOREST PLOTS -2022-05-16





Trial NCT04362137; EudraCT 2020-001662-11
Publication RUXCOVID - Han MK, Lancet Rheumatol (2022) (published paper)
Dates: 2020-05-04 to 2020-09-19
Funding: Private (Novartis and Incyte)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / Russia, USA, Brazil, Spain,
Argentina, Peru, Turkey, Mexico, UK, Colombia, France,
and Germany Follow-up duration (days): 29 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Ruxolitinib 5 mg tablets orally twice daily for 14 days with possible extension of treatment to 28 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Placebo=145 Ruxolitinib=287 | |
| Characteristics of participants N= 432 Mean age : NR 235 males Severity : Mild: n=141 / Moderate: n=268 / Severe: n=22 Critical: n=0 | |
| Primary outcome | |
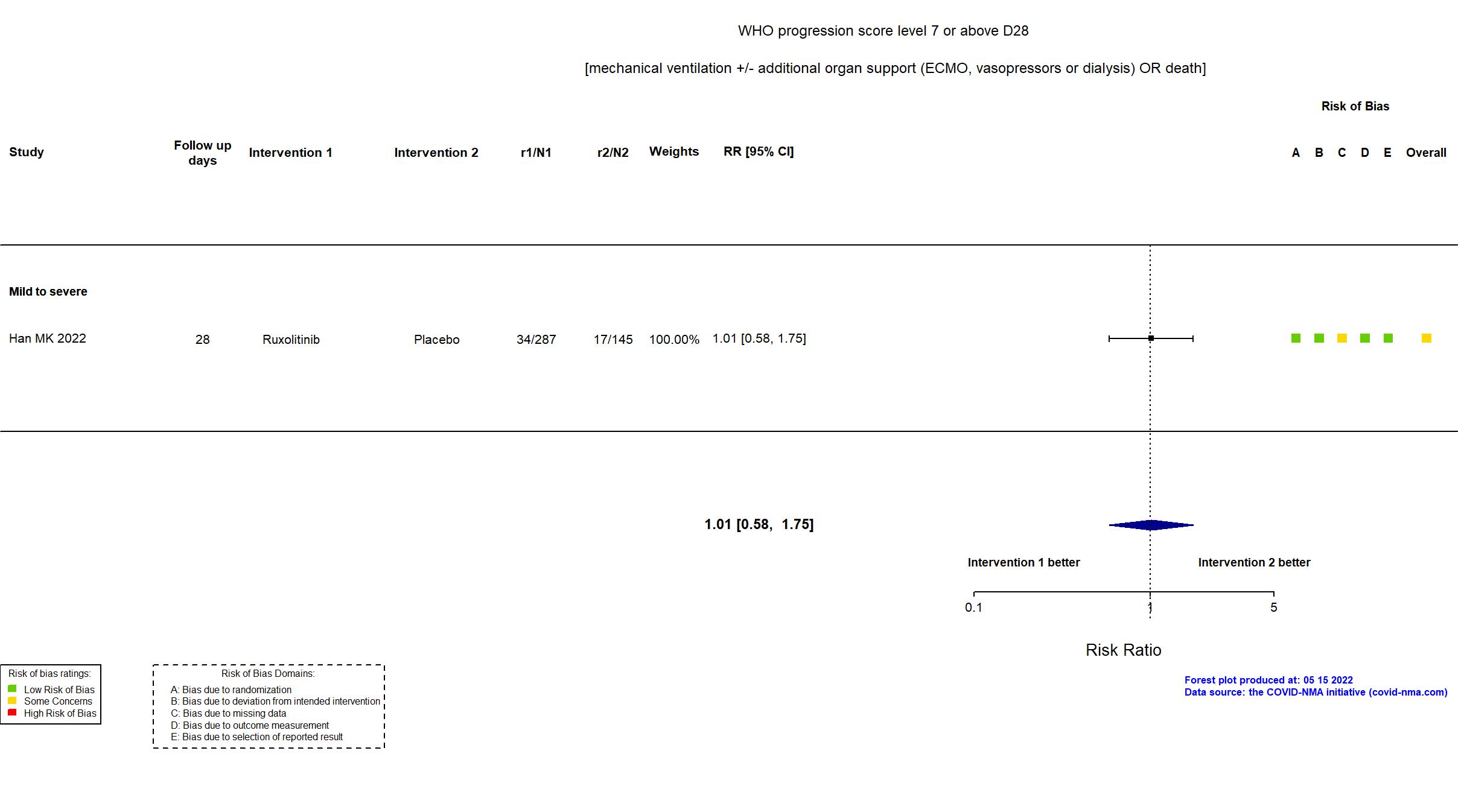
| In the register Proportion of Patients Who Die, Develop Respiratory Failure [Require Mechanical Ventilation] or Require Intensive Care Unit (ICU) Care [ Time Frame: Day 1 - Day 29 ] | |
| In the report Composite of death, respiratory failure (requiring invasive mechanical ventilation), or ICU care, by day 29 | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
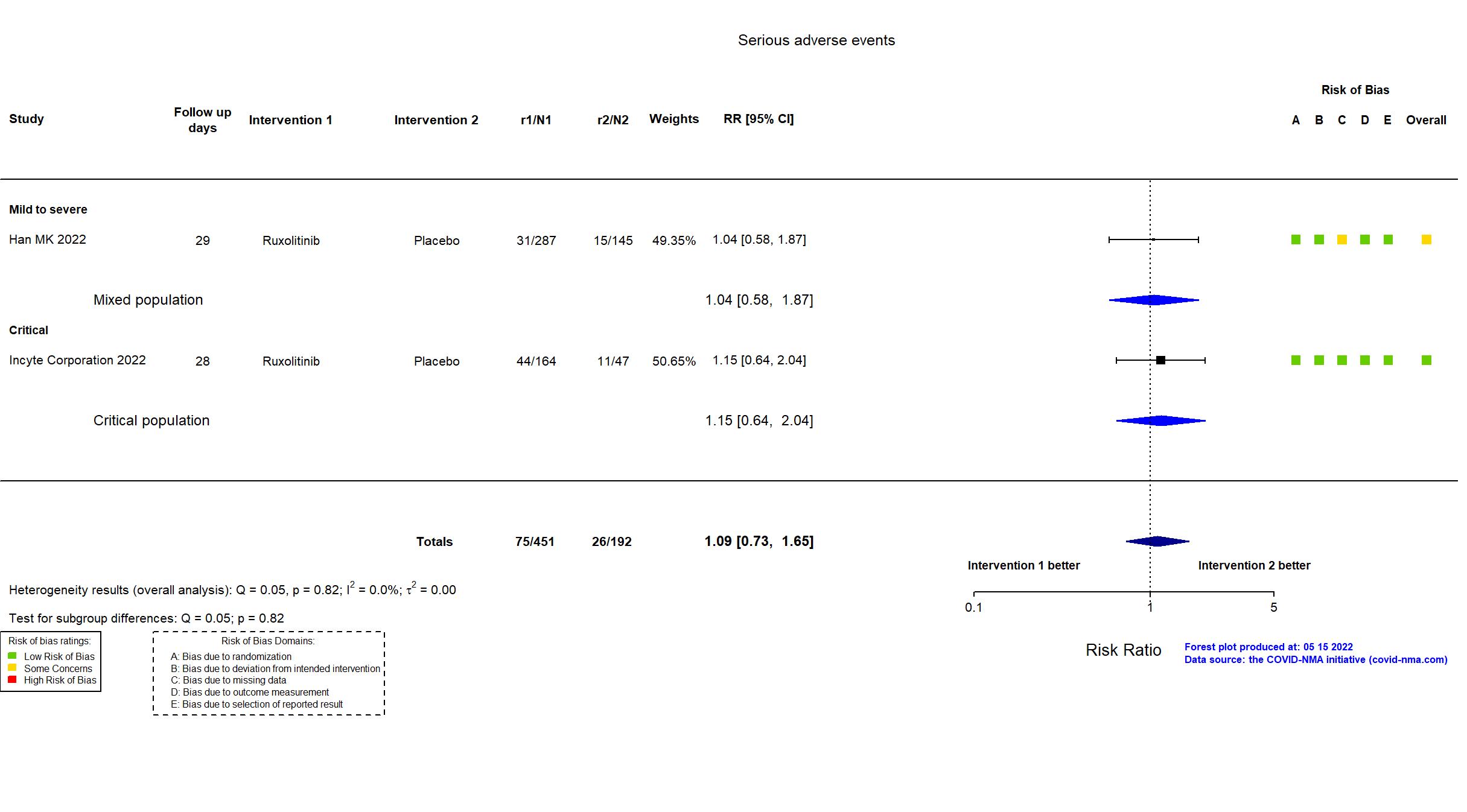
In addition to the trial registry, the study protocol, statistical analysis plan, results summary and clinical trial report were used in data extraction and assessment of risk of bias. No pre-print or published article was available. The study achieved the target sample size specified in the trial registry. There is no change from the trial registration in the intervention and control treatments. Immunosuppressants were not allowed as part of eligibility criteria but 58% of participants received steroids at baseline. The primary outcome indicated in registry reflects the primary outcome reported in the trial results summaries. The trial (n = 432) achieved the anticipated sample size in the original version of the registry (n = 402). Adverse and serious adverse events were not described in the registry but are reported in the protocol.
This study was updated on May 11th, 2022 with data extracted from the published peer-reviewed report. |
Trial NCT04377620
Publication RUXCOVID-DEVENT - RUXCOVID-DEVENT, Unpublished (2022) (preprint)
Funding: Private (Incyte Corporation)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / Russia and the USA Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Ruxolitinib 15 mg twice a day for 14 days through enteric feeding tube or orally Ruxolitinib 5mg 5 mg twice a day for 14 days through enteric feeding tube or orally Ruxolitinib 15mg 15 mg twice a day for 14 days through enteric feeding tube or orally |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Ruxolitinib =164 Placebo=47 Ruxolitinib 5mg=87 Ruxolitinib 15mg=77 | |
| Characteristics of participants N= 375 Mean age : NR 185 males Severity : Mild: n=0 / Moderate: n=0 / Severe: n=0 Critical: n=288 | |
| Primary outcome | |
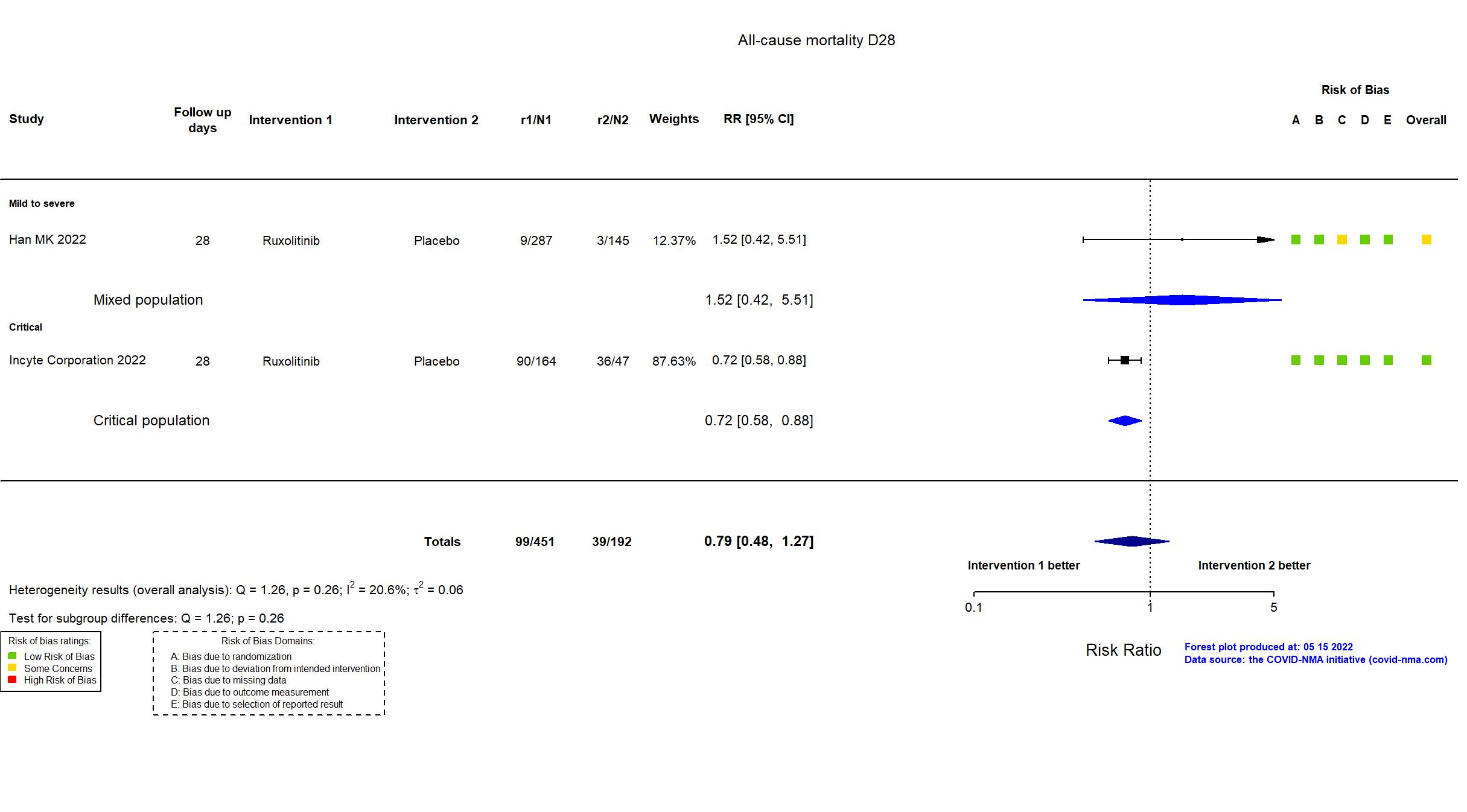
| In the register Percentage of Participants Who Have Died Due to Any Cause [ Time Frame: Study start to Day 29 ] To evaluate the 28-day mortality rate of ruxolitinib 5 mg BID + SoC therapy and ruxolitinib 15 mg BID + SoC compared with placebo + SoC therapy, in participants with COVID-19-associated ARDS who require mechanical ventilation. | |
| In the report NR | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Low |
| General comment | “In addition to the study results posted to ClinicalTrials.gov, the protocol, statistical analysis plan and prospective study registry were used in data extraction and risk of bias assessment. No manuscript was available. There is no change from the trial registration in the intervention and control treatments. The registry primary outcome reflects the reported primary outcome. The eligibility age was changed from ≥18 years to ≥12 years one week after start of recruitment. The study was terminated early by the study sponsor for unknown reasons that were not related to safety. As a result the study (n=211) did not achieve the target sample size (n=500) specified in the trial registry." |