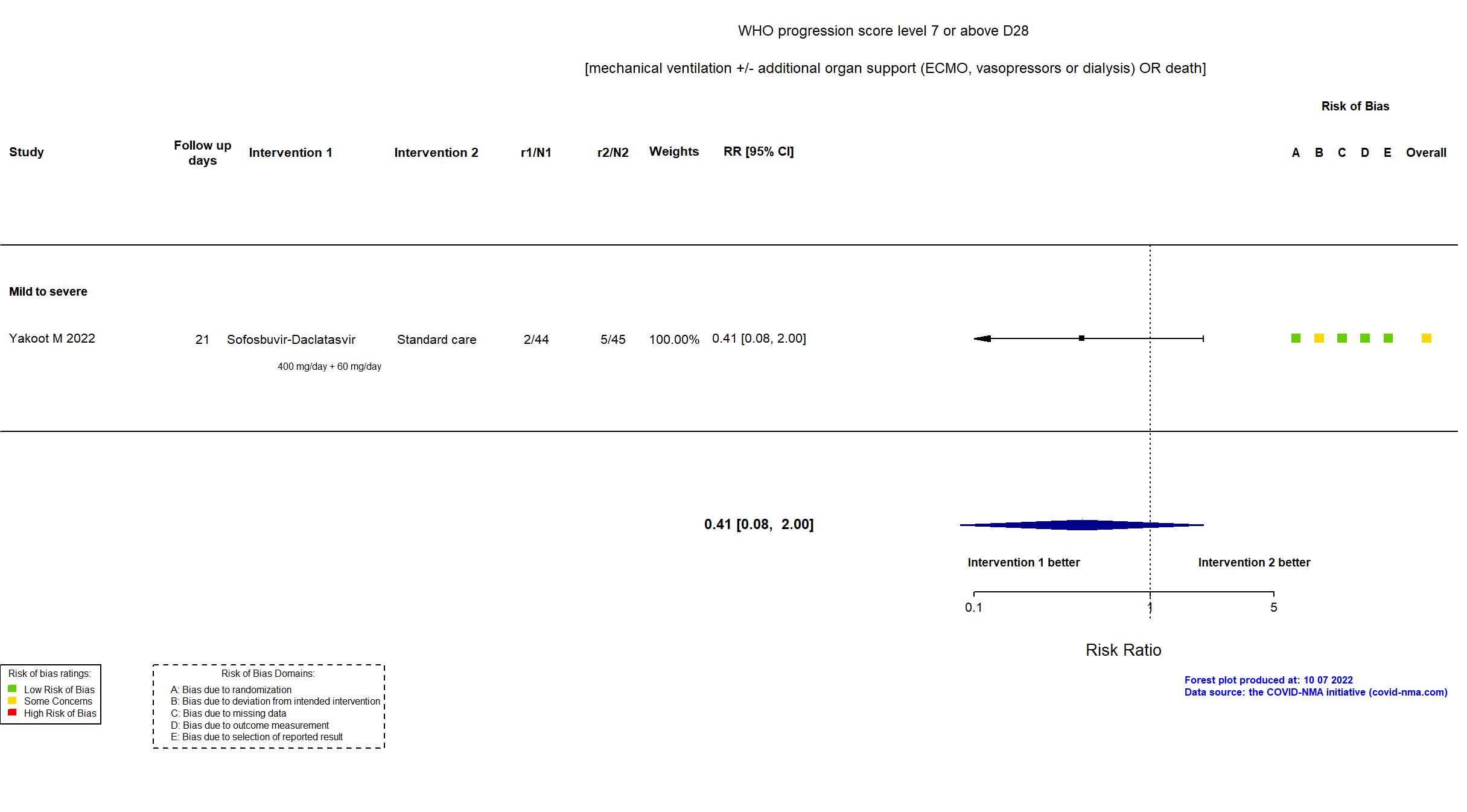
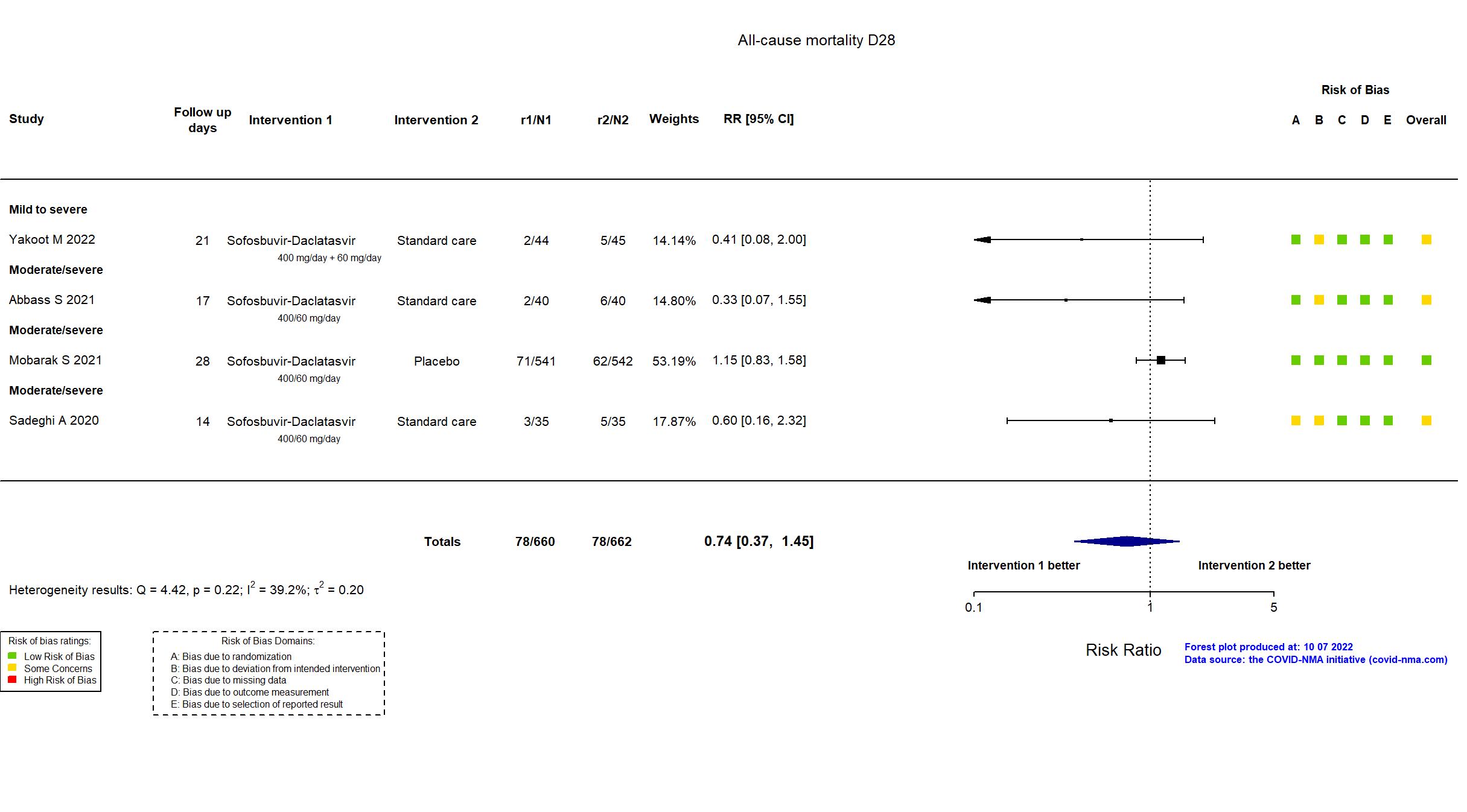
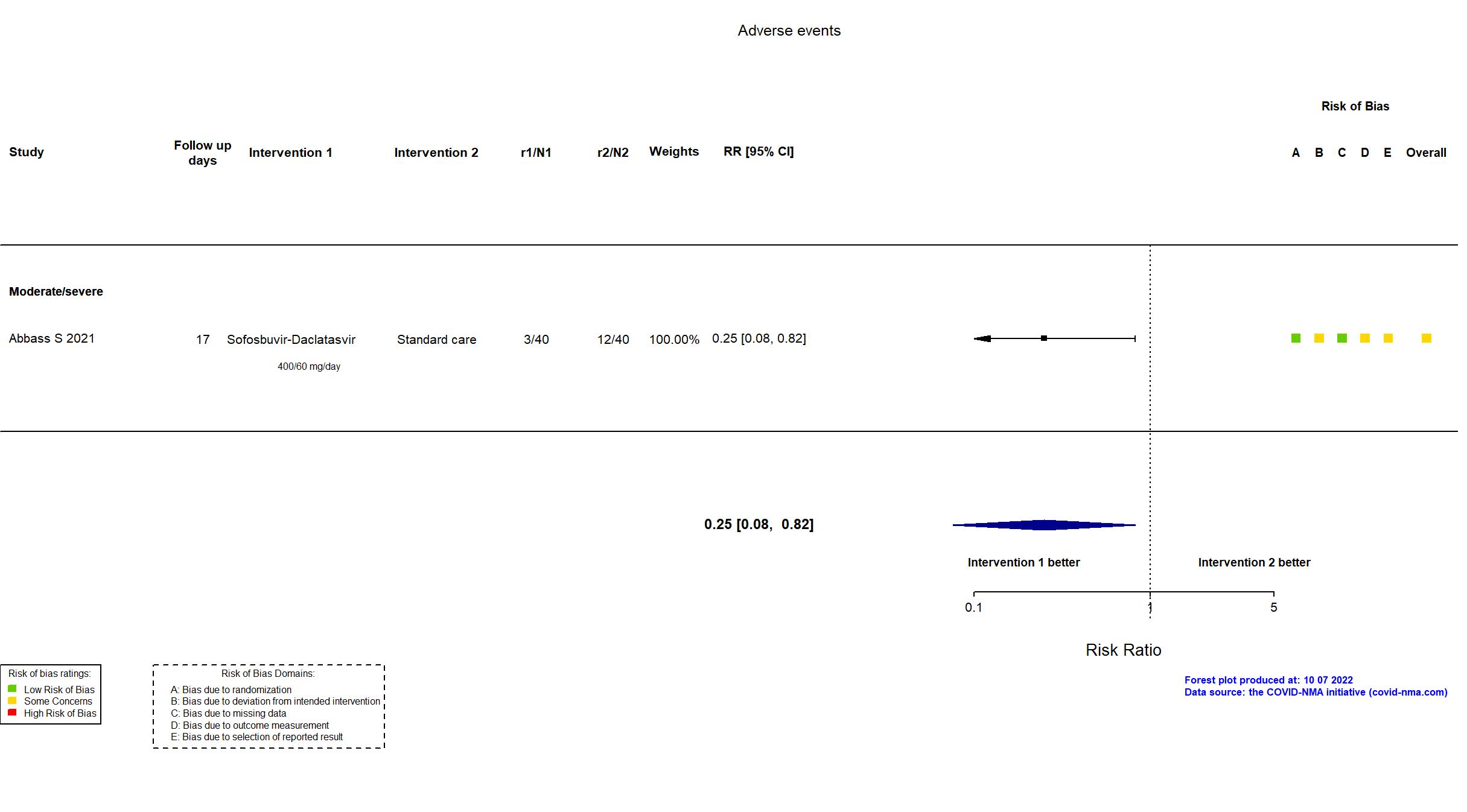
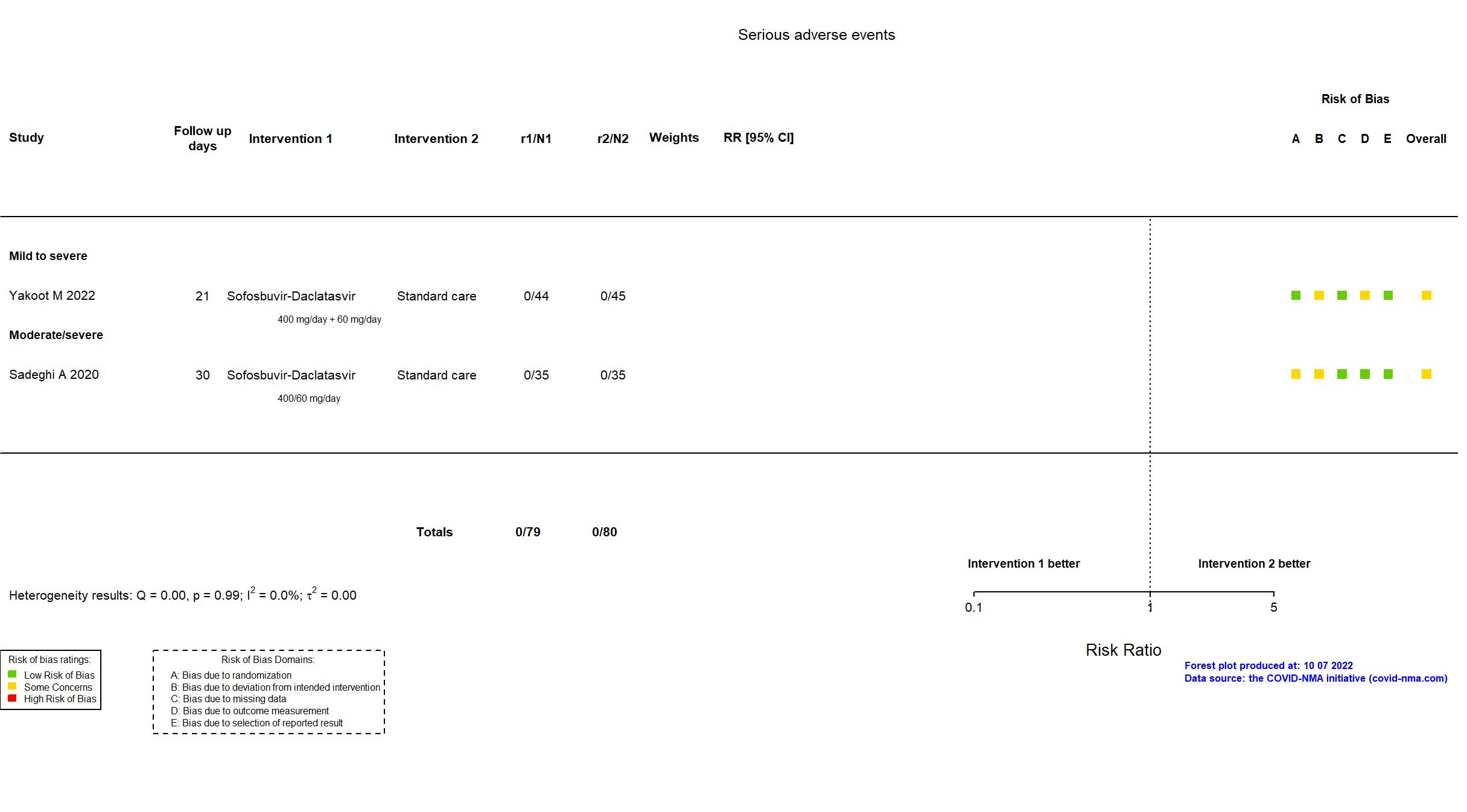
Sofosbuvir/daclatasvir vs Standard care/Placebo (RCT)
Hospitalized patients
FOREST PLOTS -2022-10-07







Trial ISRCTN21085622
Publication Abbass S, J Med Virol (2021) (published paper)
Dates: 2020-09-01 to 2021-03-30
Funding: Private (Pharco Pharmaceuticals / European Egyptian Pharmaceutical Industries (EEPI))
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / Egypt Follow-up duration (days): 17 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Sofosbuvir-Daclatasvir SOF 400 mg once a day orally + DCV 60 mg once a day orally for 10 days SOF-RDV SOF 400 mg once a day orally + RDV 200 mg once a day orally for 10 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Sofosbuvir-Daclatasvir=40 SOF-RDV=40 Standard care=40 | |
| Characteristics of participants N= 120 Mean age : NR 64 males Severity : Mild: n=0 / Moderate: n=46 / Severe: n=74 Critical: n=0 | |
| Primary outcome | |
| In the register 1. Sum of the counted symptoms (fever, headache, generalized aches (myalgia/arthralgia); respiratory distress combined with no evidence of deterioration (ICU admission and mechanical ventilation) at days3, 7 and 10; 2. Mean Oxygen saturation from day 1 to day 10 (based on daily recording as per CRF) | |
| In the report 1. Sum of the counted symptoms (fever, headache, generalized aches [myalgia/arthralgia]; respiratory distress combined with no evidence of deterioration [ICU admission and mechanical ventilation]) at Days 7 and 10, controlling for the corresponding count of symptoms at Day 3 for each patient; 2. mean change in oxygen saturation from Day 1 to Day 10 (based on daily recording per CRF) | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the study registry was used in data extraction and risk of bias assessment. Neither protocol nor statistical analysis plan was available. The primary and secondary outcomes reported in the article reflect those in the registry, which was retrospective. The study achieved the target sample size specified in the trial registry. |
Trial IRCT20200624047908N1
Publication DISCOVER - Mobarak S, J Antimicrob Chemoth (2021) (published paper)
Dates: 2020-07-13 to 2020-10-30
Funding: Public/non profit (International Treatment Preparedness coalition)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / Iran Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Sofosbuvir-Daclatasvir SOF/DCV: 400/60 mg orally once daily for 10 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Sofosbuvir-Daclatasvir=541 Placebo=542 | |
| Characteristics of participants N= 1083 Mean age : NR 585 males Severity : Mild: n=0 / Moderate: n=* / Severe: n=* Critical: n=2 | |
| Primary outcome | |
| In the register Recovery within 10 days of starting the drug. Recovery means: (no fever, no shortness of breath, no cough or improved, no fatigue or improved, tolerated oral nutrition) for 24 hours. | |
| In the report Hospital discharge within 10 days after randomization where participants were discharged on the managing physician’s decision based on the absence of fever or dyspnoea, no or improved cough and fatigue, and tolerance of oral feeding, with a stable O2 saturation of >=95%. | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published artcile, the pre-print article and the trial registry were used in data extraction and assessment of risk of bias. Neither study protocol nor statistical analysis plan was available. There were no differences between the published/pre-print article and trial registry in population, procedures and interventions. The published article reports on changes to the protocol during the study. These included broadening the inclusion criteria to allow more participants to be recruited, amending the discharge criteria and dropping the ICU outcome due to shortages of hospital beds. Admission to and length of stay in ICU, a secondary outcome in the registry, was not measured because of lack of ICU beds. The report methods state that adverse events were recorded but these were not reported. The trial achieved its pre-stated target sample size. The study was updated on December 16th, 2021 with data from the published article.. |
Trial IRCT20200128046294N2
Publication Sadeghi A, J Antimicrob Chemoth (2020) (published paper)
Dates: 26/03/2020 to 26/04/2020
Funding: Mixed (Fanavaran Rojan Mohaghegh Daru Co.; Digestive Disease Research Institute of Tehran University of Medical Sciences)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / Iran Follow-up duration (days): 30 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Sofosbuvir-Daclatasvir SOF/DCV: 400/60 mg orally once daily for 14 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Sofosbuvir-Daclatasvir=35 Standard care=35 | |
| Characteristics of participants N= 70 Mean age : NR 34 males Severity : Mild: n=0 / Moderate: n=* / Severe: n=* Critical: n=0 | |
| Primary outcome | |
| In the register Clinical recovery (composite) within 14 days from initiation of study treatment until normalization of fever (/=94% on room air), sustained for at least 24 hours | |
| In the report Clinical recovery within 14 days of enrolment | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to all available versions of the published article, the study registry was used in data extraction and risk of bias assessment. The target sample size specified in the registry was achieved. There is no change from the trial registration in the intervention and control treatments or in the primary and secondary outcomes. |
Trial DRKS00022203
Publication Yakoot M, medRxiv (2022) (preprint)
Dates: 2020-06-20 to 2020-09-30
Funding: Private (PHARCO CORPORATE)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Single center / Egypt Follow-up duration (days): 21 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Sofosbuvir-Daclatasvir 400 mg sofosbuvir + 60 mg daclatasvir orally once a day for 10 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Sofosbuvir-Daclatasvir=44 Standard care=45 | |
| Characteristics of participants N= 89 Mean age : NR 38 males Severity : Mild: n=12 / Moderate: n=61 / Severe: n=16 Critical: n=0 | |
| Primary outcome | |
| In the register 1. Proportion of clinical recovery (composite) within 14 & 21 days (Normalization of fever (≤37.2 °C oral), Respiratory rate (≤24/minute on room air), Oxygen saturation (≥94% on room air)), sustained for at least 24 hours (using Exact test with Boot-Strapping) ; 2. Time to clinical recovery (composite) [Time frame: 21 days after randomization (using Kaplan Meier curve and Cox-regression) ; 3. Time to viral negativity assessed twice 48 hours apart (log10 viral load assessed by reverse transcription-PCR) during hospital stay (Kaplan Meier curve and Cox-regression) ; 4. Mean change in Clinical status using 8 point ordinal scale [Day 3 through Day 14, 21 (using Repeated measure Factorial ANOVA test]: 1) Death; 2) Hospitalized, on invasive ventilation or ECMO; 3) Hospitalized, on NIV or HFO2; 4) Hospitalized with oxygen supplement; 5) Hospitalized, not requiring oxygen but need medical care; 6) Hospitalized, not requiring supplemental oxygen or ongoing medical care; 7) Not hospitalized, limitation on activities and/or requiring home oxygen; 8) Not hospitalized with normal activity. | |
| In the report 1) Proportions-of and 2) time-to-clinical recovery within the 21day time-frame following enrolment | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the pre-print article, the trial registry, protocol, statistical analysis plan and supplementary appendices were used in data extraction and assessment of risk of bias. In the registry the study had three arms (sofosbuvir/daclatasvir + SOC, hydroxychloroquine + SOC, and SOC alone), but the article reports on two arms (sofosbuvir/daclatasvir + SOC, and SOC alone, with both arms receiving hydroxychloroquine as part of SOC). The primary outcome indicated in registry reflects the primary outcome reported in the paper. |