Interferon alpha2b vs Standard care (RCT)
Hospitalized patients
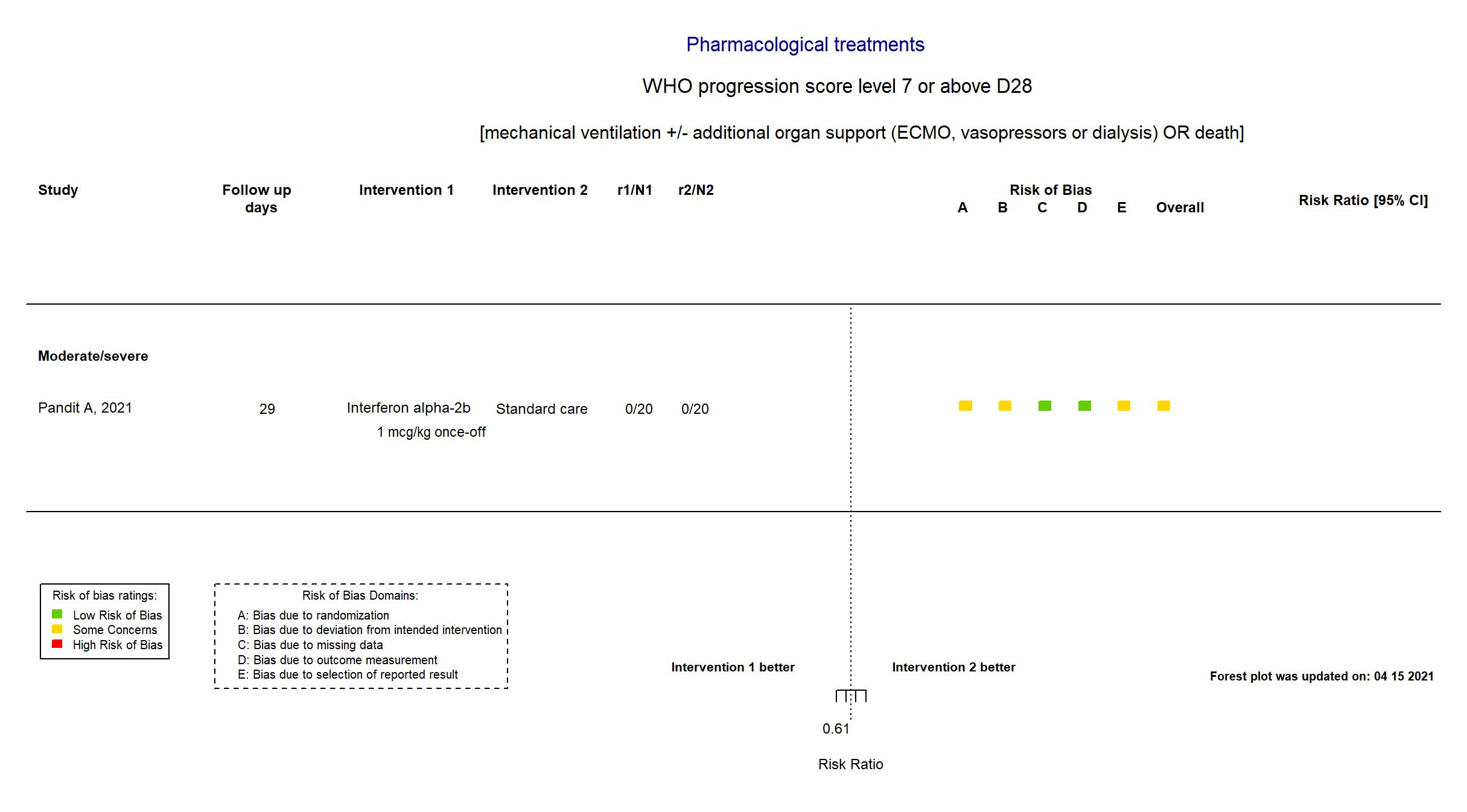
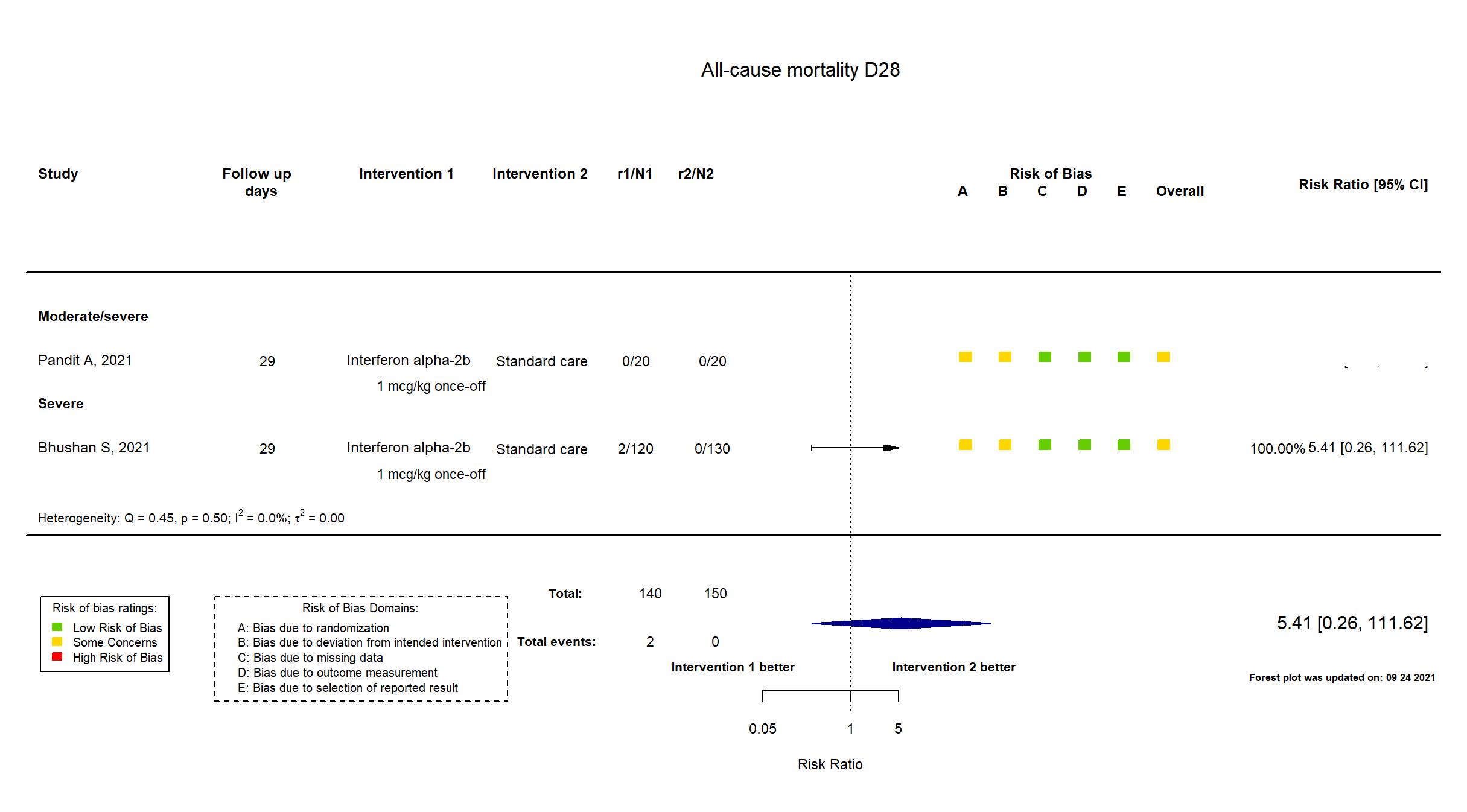
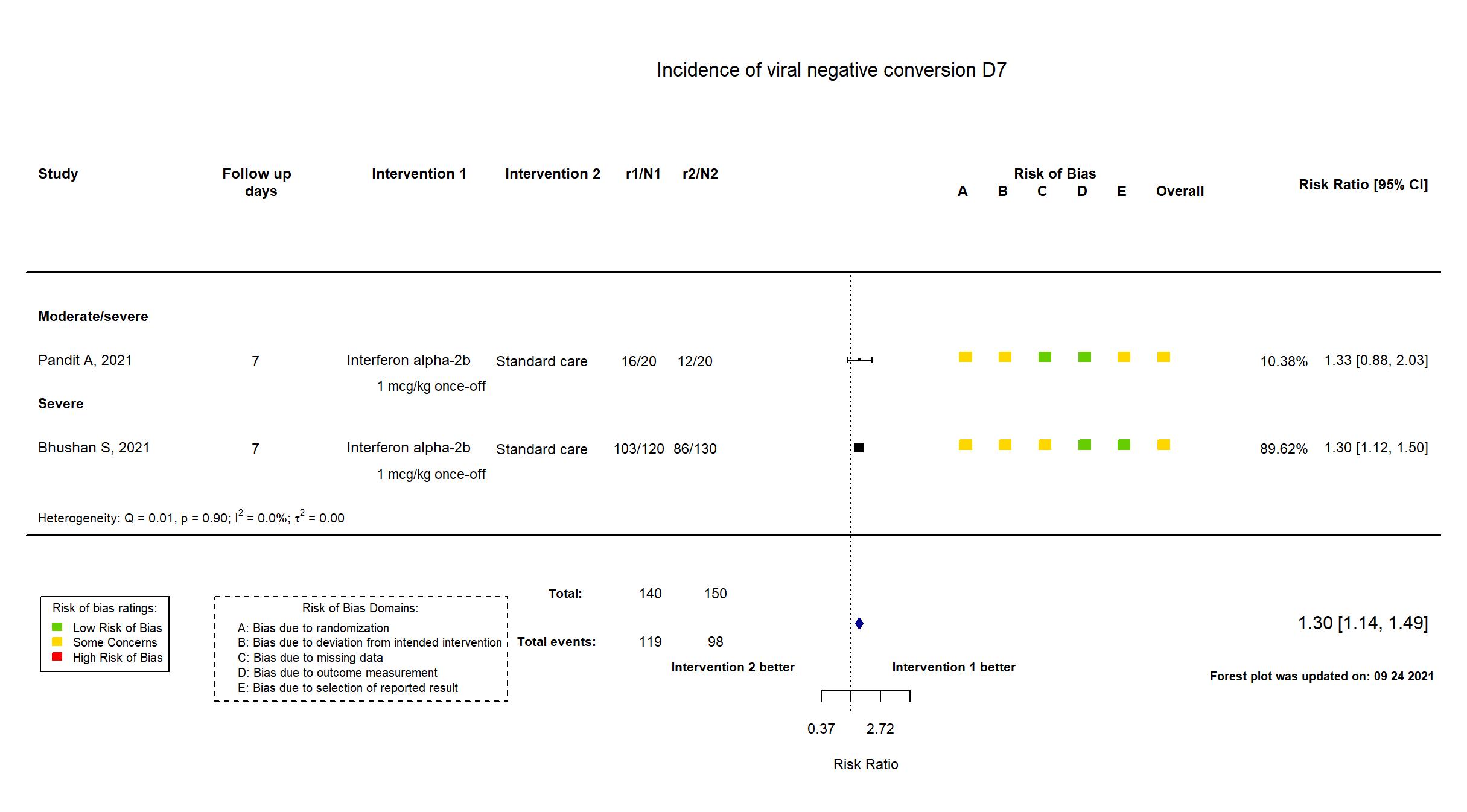
FOREST PLOTS -2022-03-04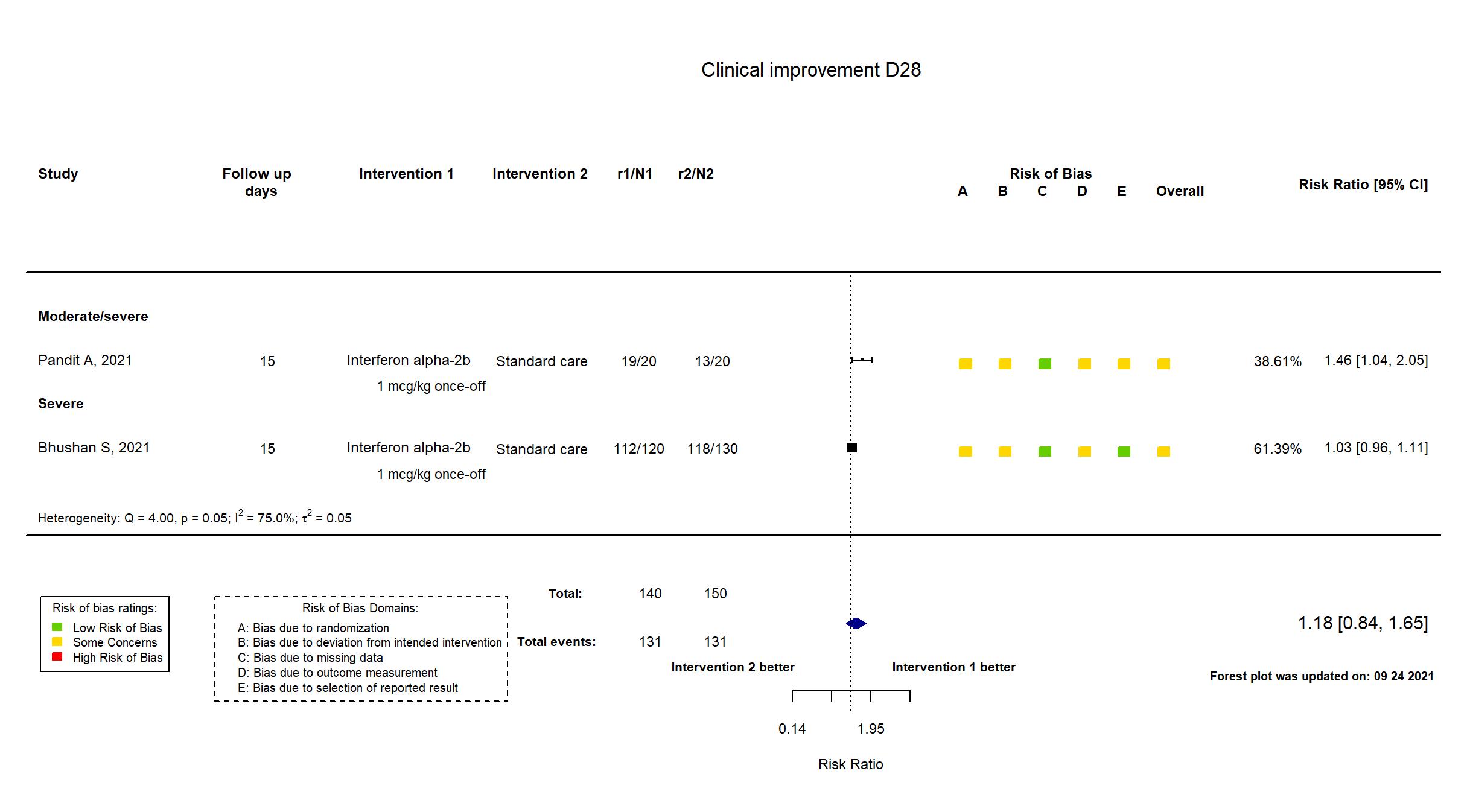
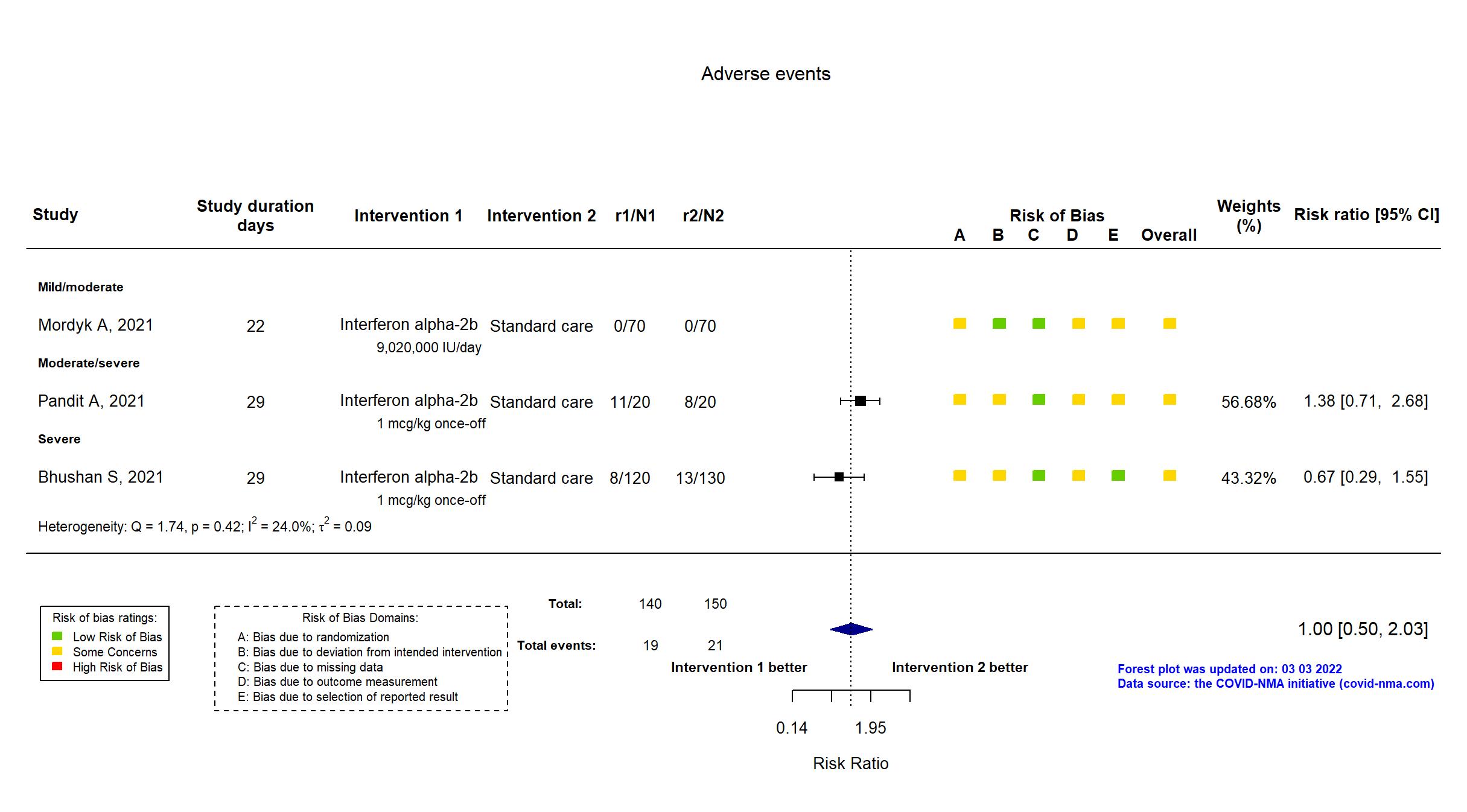
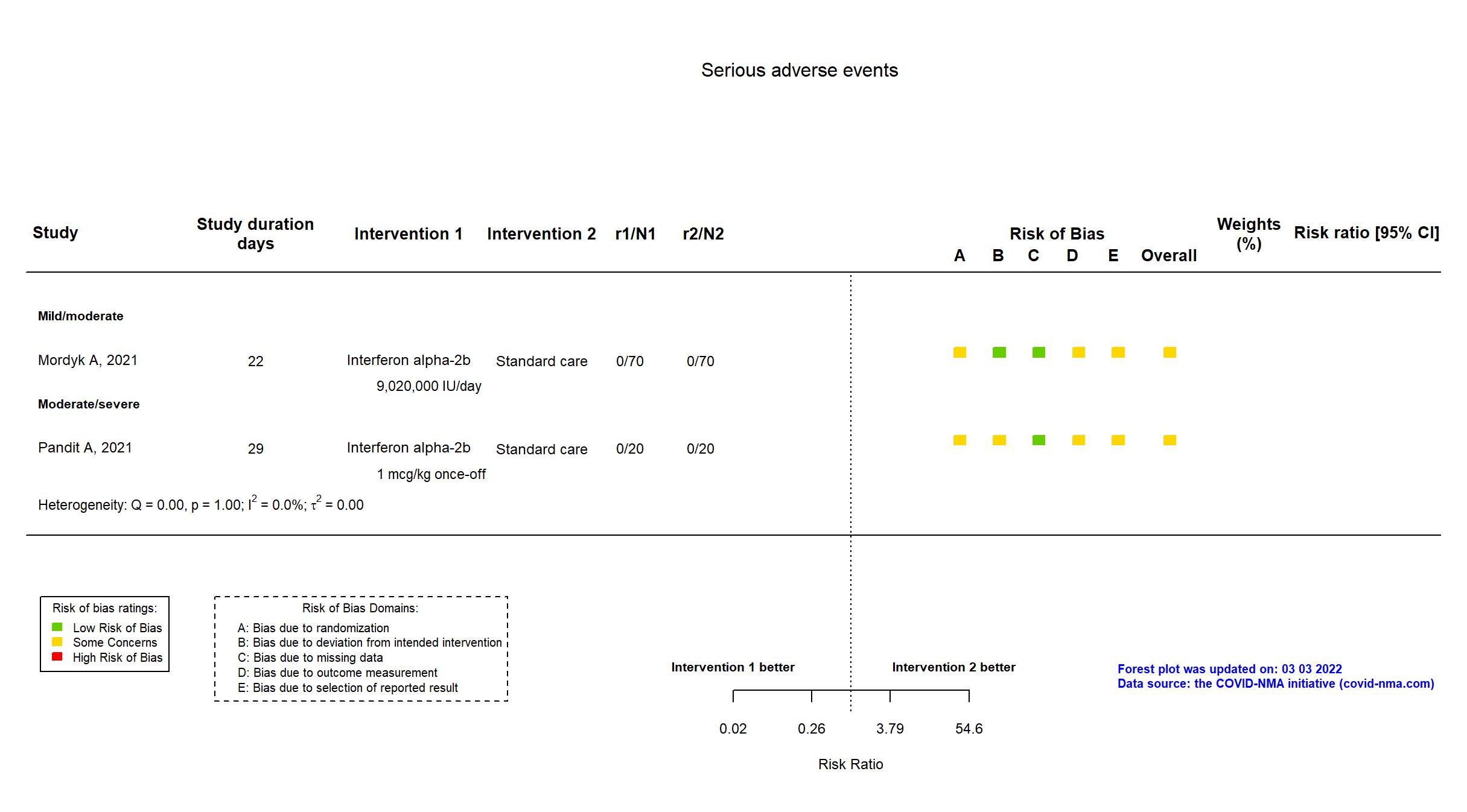






Trial CTRI/2020/12/029855
Publication Bhushan S, Int J Infect Dis (2021) (published paper)
Dates: 2020-12-16 to 2021-03-25
Funding: Private (Cadila Healthcare Ltd.)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / India Follow-up duration (days): 29 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Interferon alpha-2b 1 mcg/kg subcutaneously once-off |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Interferon alpha-2b=120 Standard care=130 | |
| Characteristics of participants N= 250 Mean age : NR 177 males Severity : Mild: n=0 / Moderate: n=0 / Severe: n=250 Critical: n=0 | |
| Primary outcome | |
| In the register Change in ordinal scale at Day 11 ± 1. | |
| In the report Change in ordinal scale at day 11 (two-point improvement in the WHO 7-point ordinal scale) | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the prospective trial registry and supplementary materials were used in data extraction and assessment of risk of bias. Neither protocol nor statistical analysis plan was available. The study achieved its target sample size. There is no change from the trial registration in the intervention and control treatments. The primary outcome reported reflects that in the registry. SAE was an outcome in the registry; two deaths are reported in the safety outcomes section, but it is not clear whether these were the only SAEs. Other than that, the secondary outcomes reported reflect those in the registry. Four of five risk of bias domains were assessed to have some concerns. Commonly, this would be a reason to elevate the study overall to high risk of bias, but no individual outcome was some concerns in more than three domains. |
Trial *
Publication Mordyk A, Infektsionnye Bolezni (2021) (published paper)
Funding: No specific funding
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / Russia Follow-up duration (days): 22 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Interferon alpha-2b 3,000,000 IU rectally 3 times a day for 14 days; 4000 IU topically (nasal mucosa and palatine tonsils) 5 times a day for 14 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Interferon alpha-2b=70 Standard care=70 | |
| Characteristics of participants N= 140 Mean age : NR 70 males Severity : Mild: n=* / Moderate: n=* / Severe: n=0 Critical: n=0 | |
| Primary outcome | |
| In the register NR | |
| In the report NR | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | Only the published article was used in data extraction and risk of bias assessment. The abstract was available in English while the full paper was translated from Russian using Google translate. Neither the protocol, statistical analysis plan or registry were available and therefore we cannot determine if the intervention and outcomes were determined a priori. |
Trial CTRI/2020/06/026087
Publication Pandit A, Int J Infect Dis (2021) (published paper)
Dates: 2020-07-08 to 2020-09-04
Funding: Private (Cadila Healthcare Limited)
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / India Follow-up duration (days): 29 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Interferon alpha-2b 1 mcg/kg subcutaneous injection, single dose |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Interferon alpha-2b=20 Standard care=20 | |
| Characteristics of participants N= 40 Mean age : NR 30 males Severity : Mild: n=0 / Moderate: n=* / Severe: n=* Critical: n=0 | |
| Primary outcome | |
| In the register Evaluation of the clinical efficacy of Pegylated IFN-?2b on the basis of change in ordinal scale | |
| In the report Clinical status assessed on day 15 on a WHO 7-point ordinal scale consisting of the following categories: 1, not hospitalized, no limitations of activities; 2, not hospitalized, limitation on activities; 3, hospitalized, not requiring supplemental oxygen; 4, hospitalized, requiring supplemental oxygen; 5, hospitalized, on non-invasive ventilation or high flow oxygen devices; 6, hospitalized, on invasive mechanical ventilation or extracorporeal membrane oxygenation (ECMO); and 7, death | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the study registry was used in data extraction and risk of bias assessment. No study protocol or statistical analysis plan was available. The study achieved its prespecified target sample size. Some secondary outcomes were not pre-specified including occurrence and duration of supplemental oxygen and mechanical ventilation, duration of hospitalization, change from baseline in white blood cell count (WBC), hemoglobin (Hb), platelets, creatinine, glucose, total bilirubin, ALT and AST, and change from baseline in CRP, IL-6, D-dimer, interferon-g, ferritin, TNF-α, and IL-1β until Day 14. The primary outcome (i.e., clinical improvement) was only reported at one of two pre-specified timepoints (i.e., reported at day 15 but not day 30). The published article specifies administration of a single dose of the intervention which differs from the two doses (day 1 and day 8) specified in the registry. |