Methylprednisolone vs Dexamethasone (RCT)
Hospitalized patients
Studies included but not extracted/included in the analysis: Salton F, Eur Respir J, 2022; Corral-Gudino L, Eur J Clin Invest, 2022
FOREST PLOTS -2022-08-04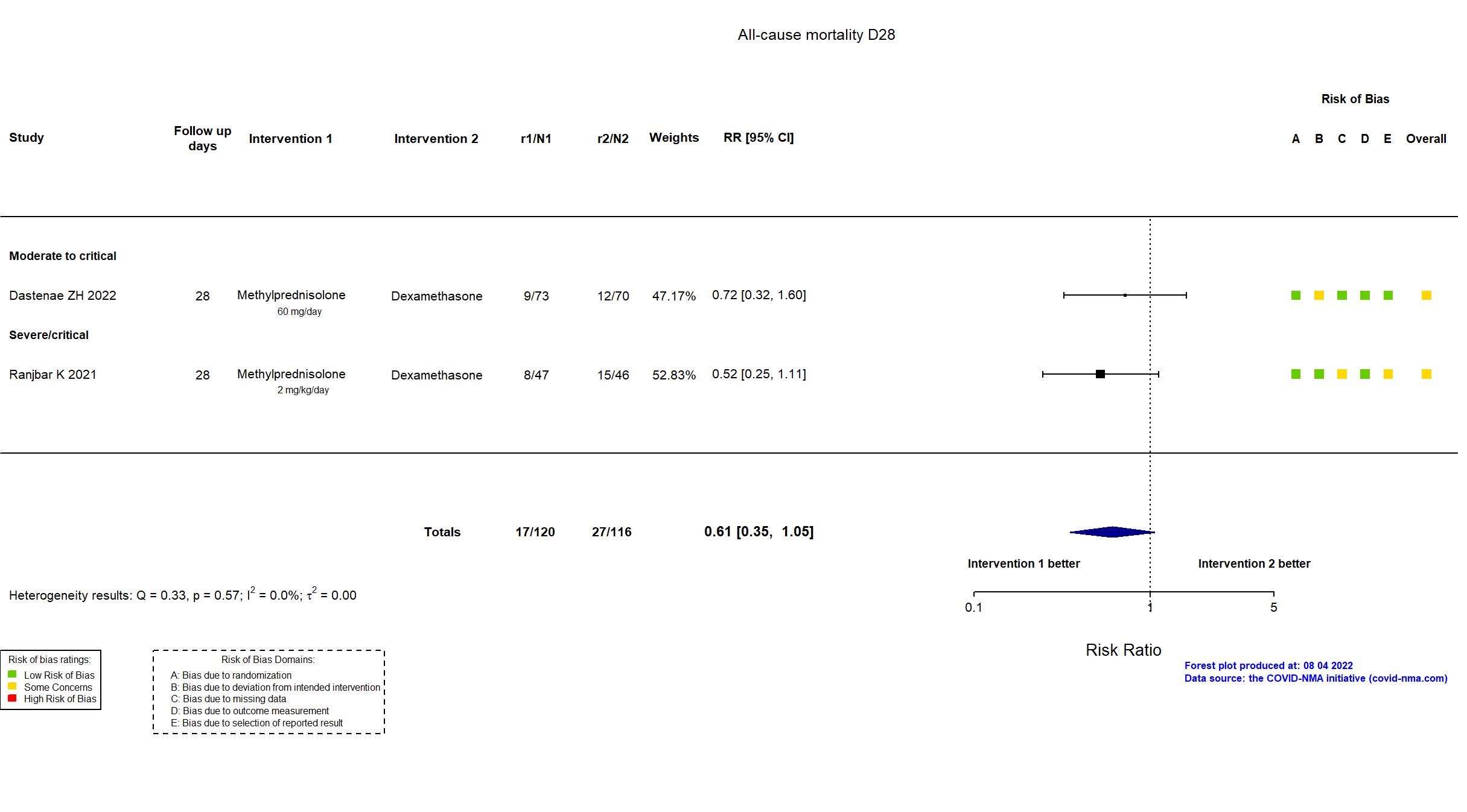
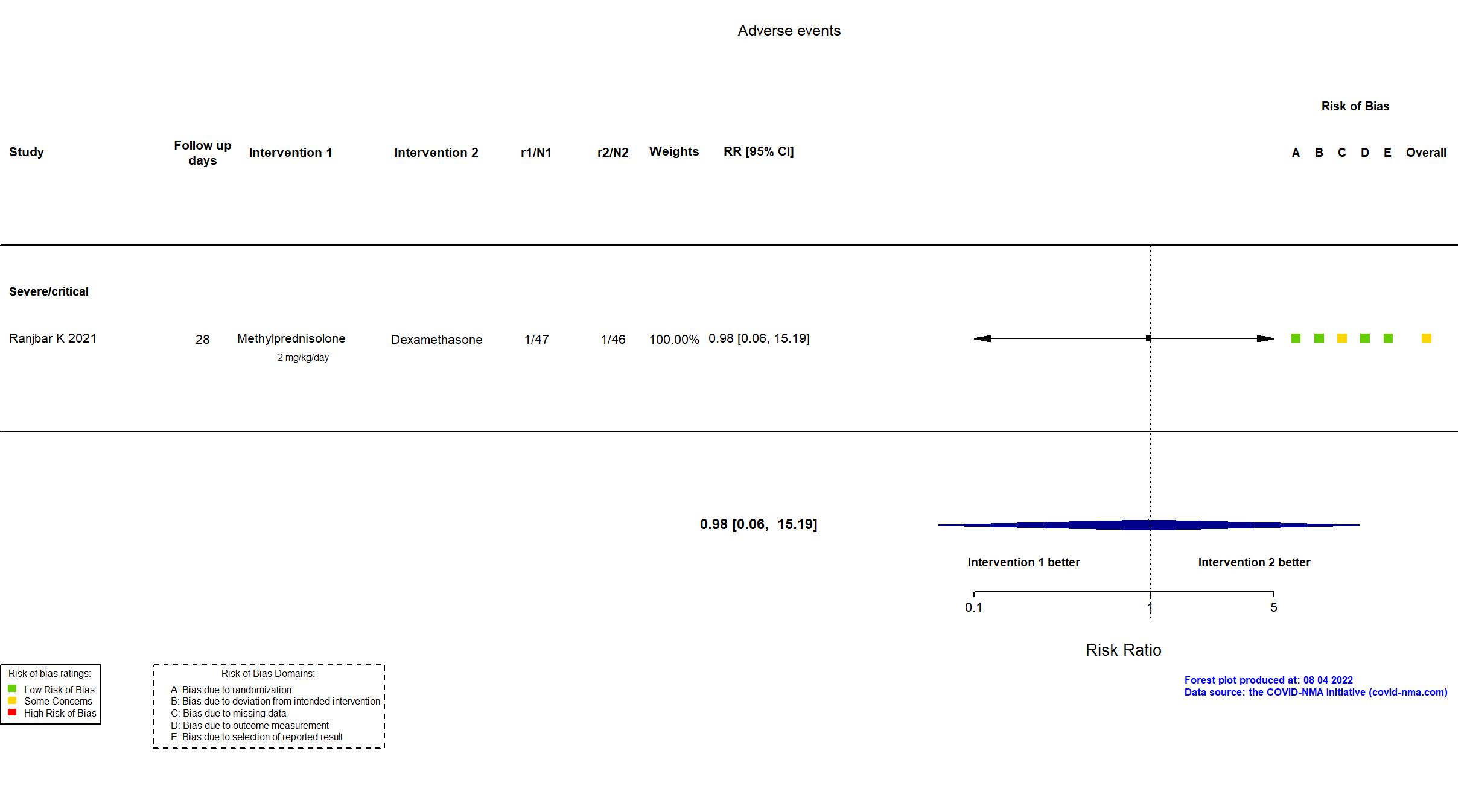
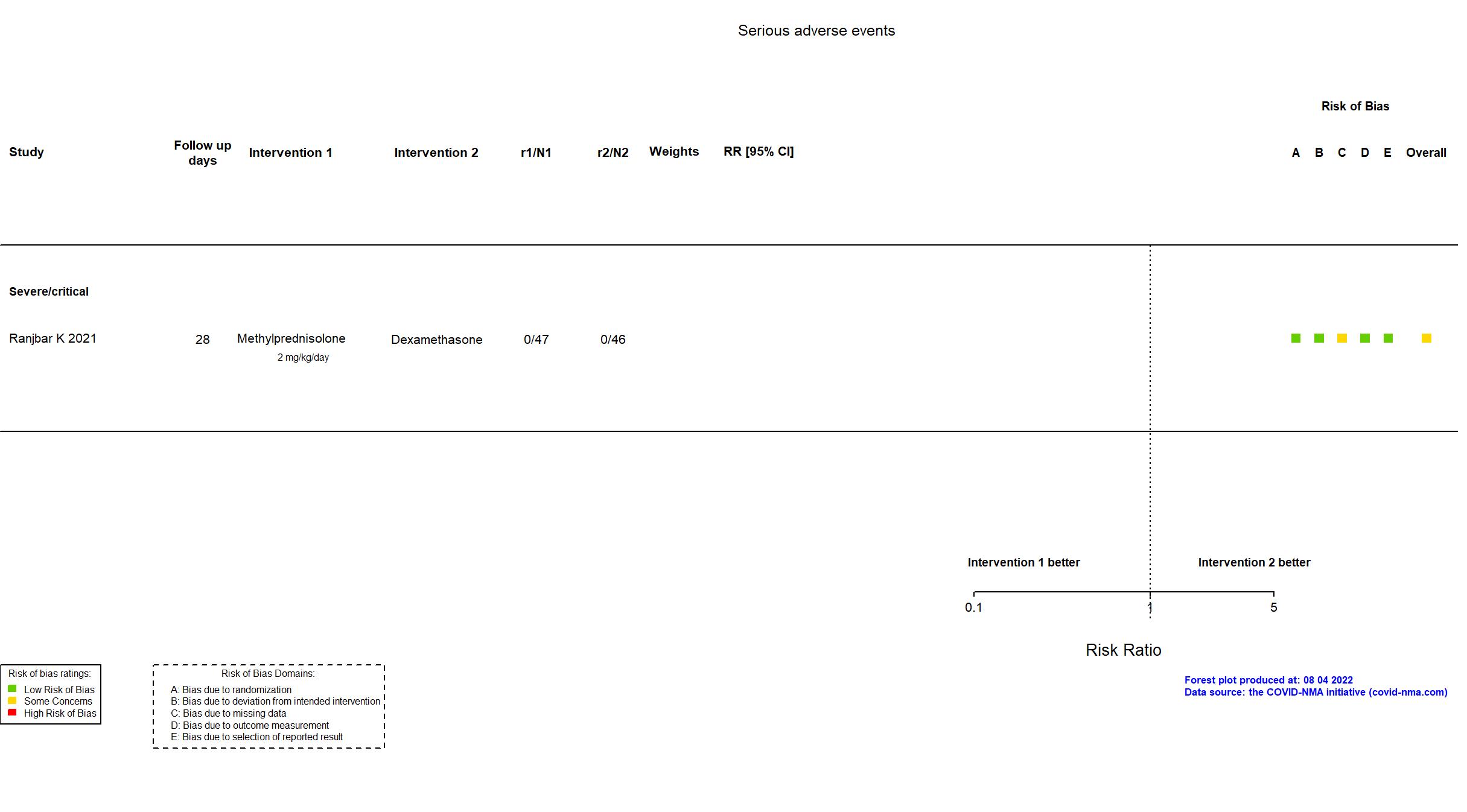
Studies included but not extracted/included in the analysis: Salton F, Eur Respir J, 2022; Corral-Gudino L, Eur J Clin Invest, 2022
FOREST PLOTS -2022-08-04


Trial IRCT20210223050466N1
Publication Dastenae ZH, Int J Infect Dis (2022) (published paper)
Dates: 2021-04-01 to 2021-06-30
Funding: Public/non profit (Shahrekord University of Medical Sciences)
Conflict of interest: No
| Methods | |
| RCT Blinding: single blinding | |
| Location :
Single center / Iran Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Methylprednisolone 60 mg intravenously per day for up to 10 days (2/3 of the dose in the morning and 1/3 in the afternoon) |
|
| Control
Dexamethasone 8 mg intravenously per day for up to 10 days (2/3 of the dose in the morning and 1/3 in the afternoon) | |
| Participants | |
| Randomized participants : Dexamethasone=70 Methylprednisolone=73 | |
| Characteristics of participants N= 143 Mean age : NR 80 males Severity : Mild: n=0 / Moderate: n=* / Severe: n=* Critical: n=* | |
| Primary outcome | |
| In the register Duration of hospitalization [Timepoint: From admission to discharge] | |
| In the report Duration of hospitalization | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the trial registry was used in data extraction and assessment of risk of bias. Neither protocol nor statistical analysis plan was available. There is no change from the trial registration in the intervention and control treatments. The primary outcome in the article reflects that in the registry. The trial (n = 143) achieved its target sample size (n = 140). All adverse events were not reported. Baseline severity and oxygen therapies in each group were not well described. |
Trial IRCT20200204046369N1
Publication Ranjbar K, BMC Infect Dis (2021) (published paper)
Dates: 2020-08-10 to 2020-11-15
Funding: Public/non profit (Research deputy chancellor of Shiraz university of medical sciences)
Conflict of interest: No
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Single center / Iran Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Methylprednisolone 2 mg/kg IV once daily over 60 minutes for 10 days, tapered to half dosage after 5 days |
|
| Control
Dexamethasone 6 mg IV once daily for 10 days | |
| Participants | |
| Randomized participants : Methylprednisolone=47 Dexamethasone=46 | |
| Characteristics of participants N= 93 Mean age : NR 49 males Severity : Mild: n=0 / Moderate: n=0 / Severe: n=* Critical: n=* | |
| Primary outcome | |
| In the register The all-cause mortality in 28 days and Clinical status | |
| In the report All-cause mortality in 28 days; Clinical status after 5 and 10 days after enrollment with 9-point WHO ordinal scale | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication:
|
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the pre-print article, the trial registry was used in data extraction and risk of bias assessment. Significant changes were made to the trial registration after the end of the study recruitment: it was changed from an unblinded, non-randomized study to a triple blinded, randomized trial; inclusion criteria 'O2 saturation of less than 92 in room air' and 'Hospitalized patients' were added; the methylprednisolone regimen was changed; the comparator was changed from standard care to dexamethasone; the sample size was increased; the primary and secondary outcome measures were changed from PAO2/fio2 (primary) and oxygen saturation, CT scan and clinical follow-up (secondary) to all-cause mortality and clinical status (primary) and mechanical ventilation, ICU admission and length of hospital stay (secondary).
On 19th of April, 2021, this study was updated based on the published report. On April 09th, 2021, we received additional information from authors on this study. This study was updated with data from contact with authors on April 19th, 2021. |