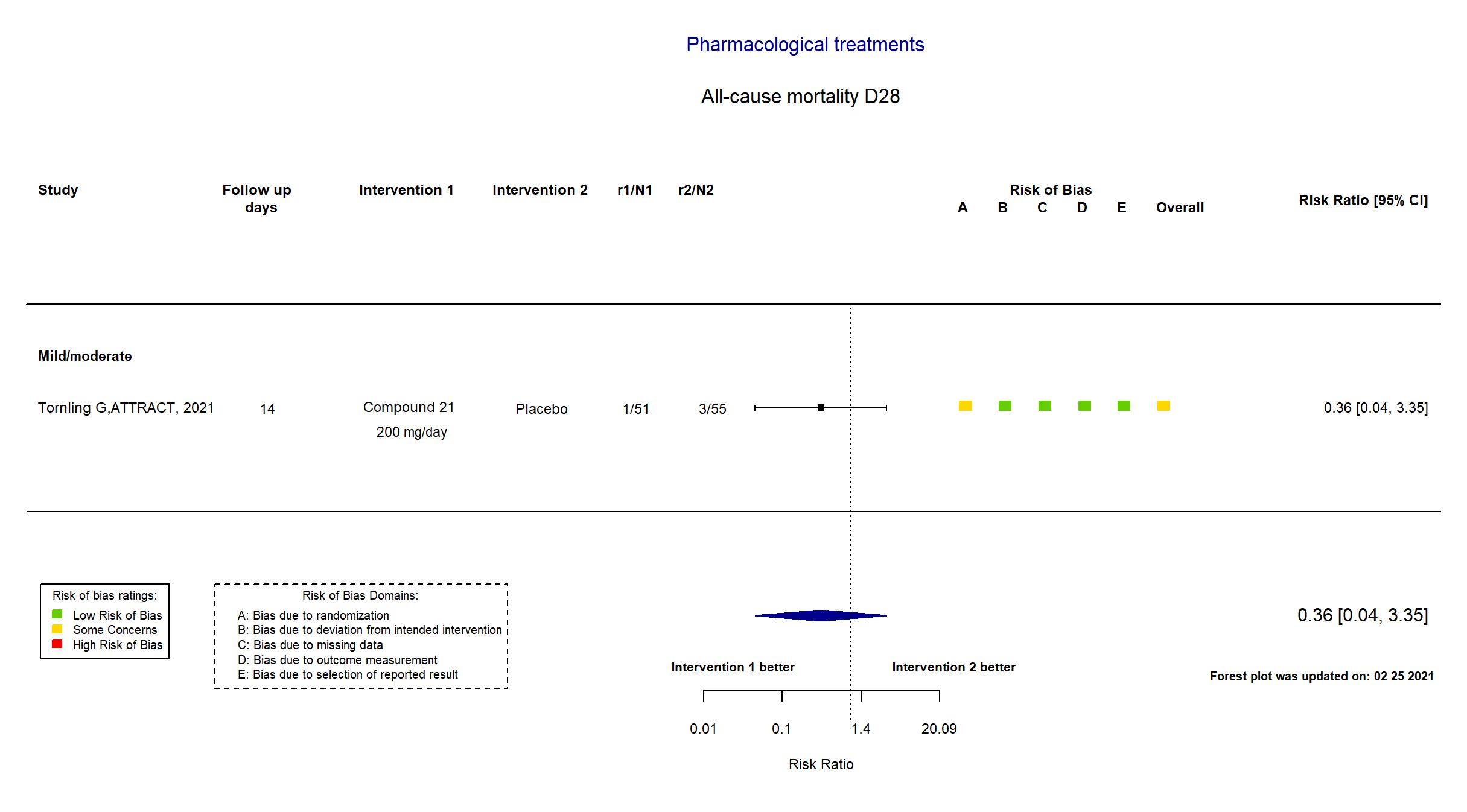
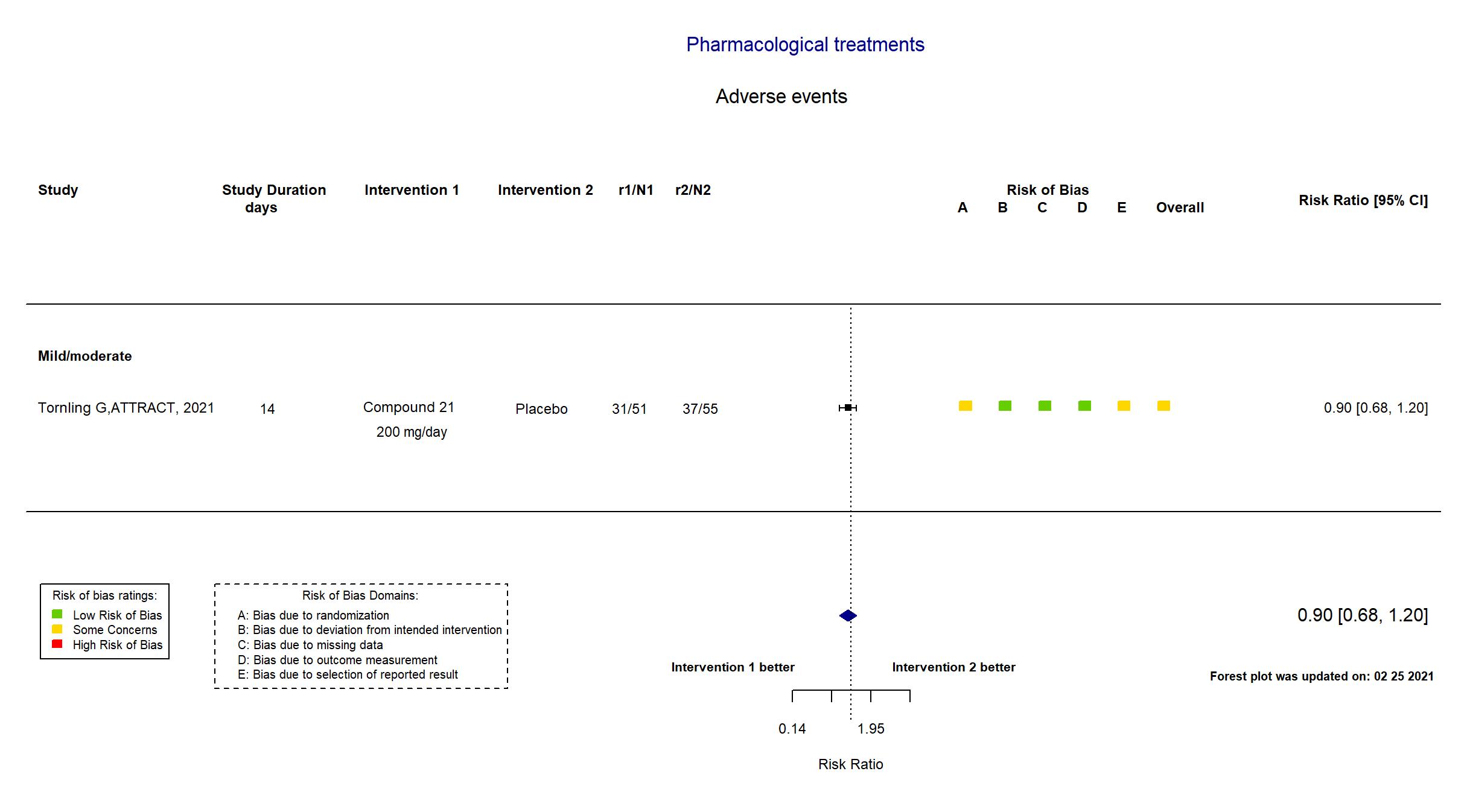
Compound 21 vs Placebo (RCT)
Hospitalized patients
FOREST PLOTS -2021-02-25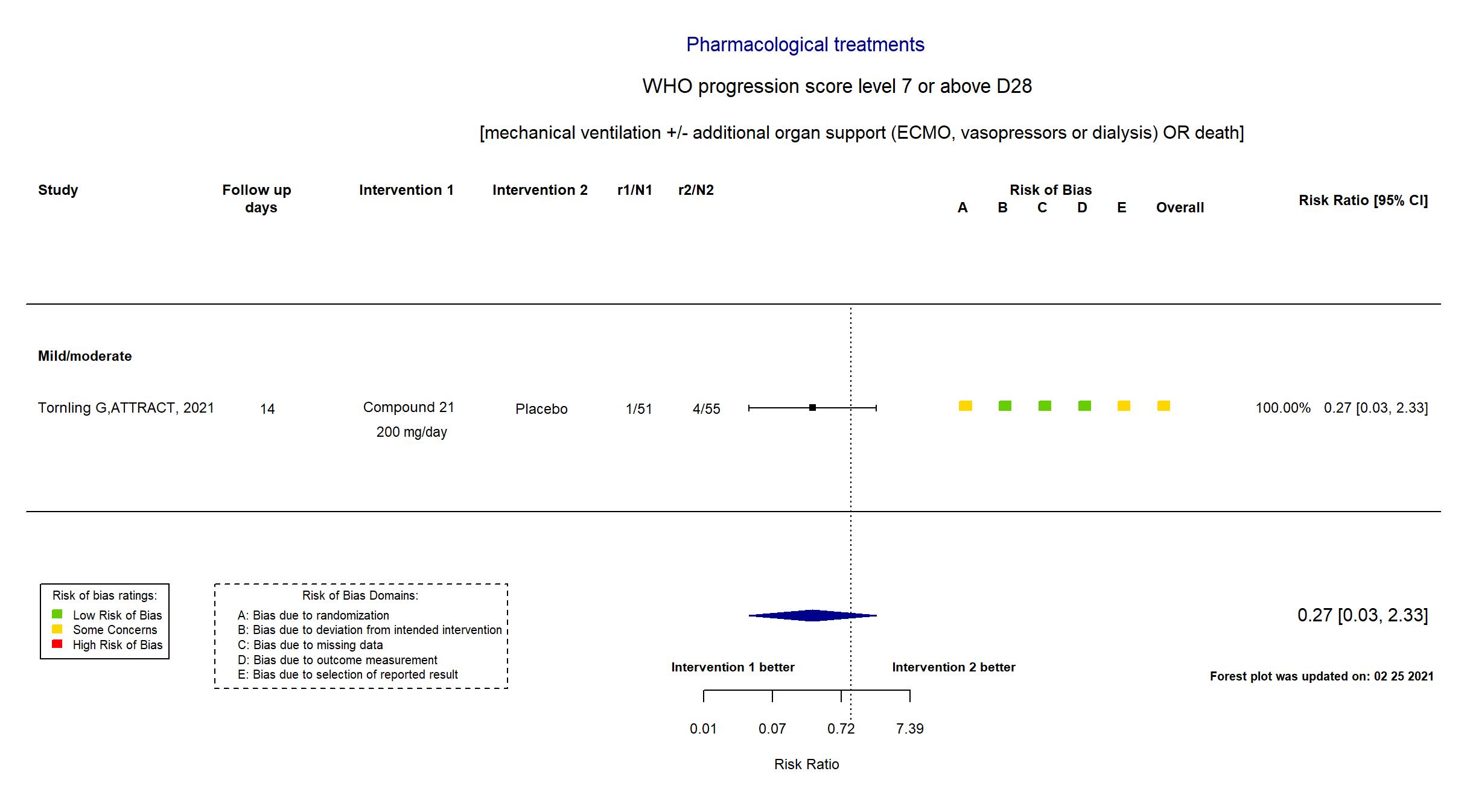



Trial NCT04452435
Publication ATTRACT - Tornling G, EClinicalMedicine (2021) (published paper)
Dates: 2020-07-21 to 2020-09-29
Funding: Mixed (Vicore Pharma; LifeArc)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: quadruple blinding | |
| Location :
Multicenter / India Follow-up duration (days): 14 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Compound 21 100 mg twice daily orally for 7 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Compound 21=51 Placebo=55 | |
| Characteristics of participants N= 106 Mean age : NR 80 males Severity : Mild: n=26 / Moderate: n=76 / Severe: n=0 Critical: n=0 | |
| Primary outcome | |
| In the register Change from baseline in C-reactive protein (CRP) after treatment with C21 200 mg daily dose (100 mg b.i.d.) [ Time Frame: Treatment period of 7 days ] | |
| In the report Change in CRP from baseline to end-of-treatment | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the published article and all versions of the pre-print article, the study registry (dated 30 June, 2020), protocol, and statistical analysis plan were used in data extraction and risk of bias assessment. The target sample size specified in the registry and protocol was not achieved due to recruitment difficulties. There were no substantive differences between the trial registry, study protocol and statistical analysis plan and the pre-print article. The outcomes WHO score 7 and above, Adverse events and Serious adverse events were not pre-specified.
On the 29th of July, 2021, this study was updated based on updated pre-print. On the 17th of November, the study was updated based on the published article. |