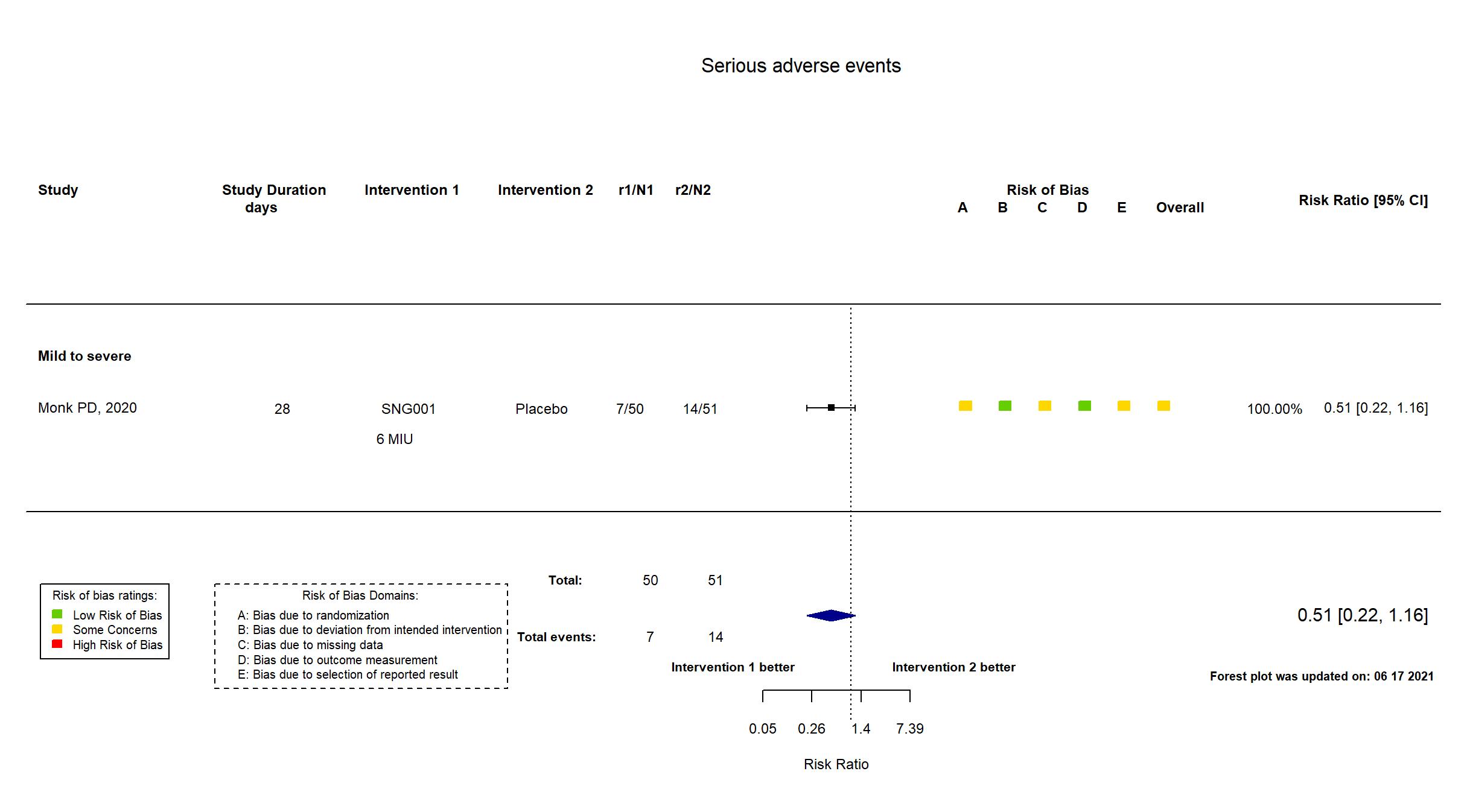
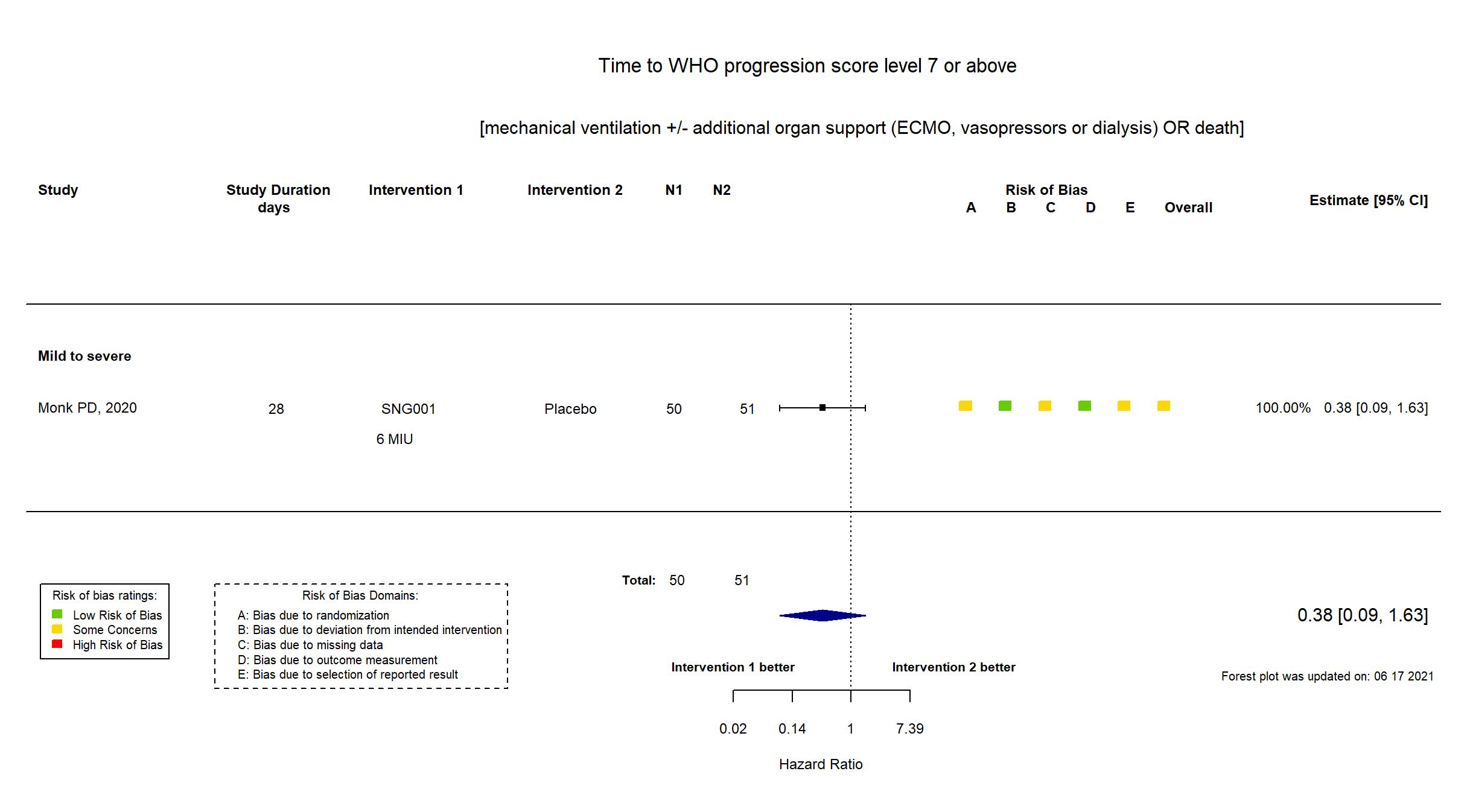
Inhaled nebulised interferon beta-1a vs Placebo (RCT)
Hospitalized patients
FOREST PLOTS -2021-06-18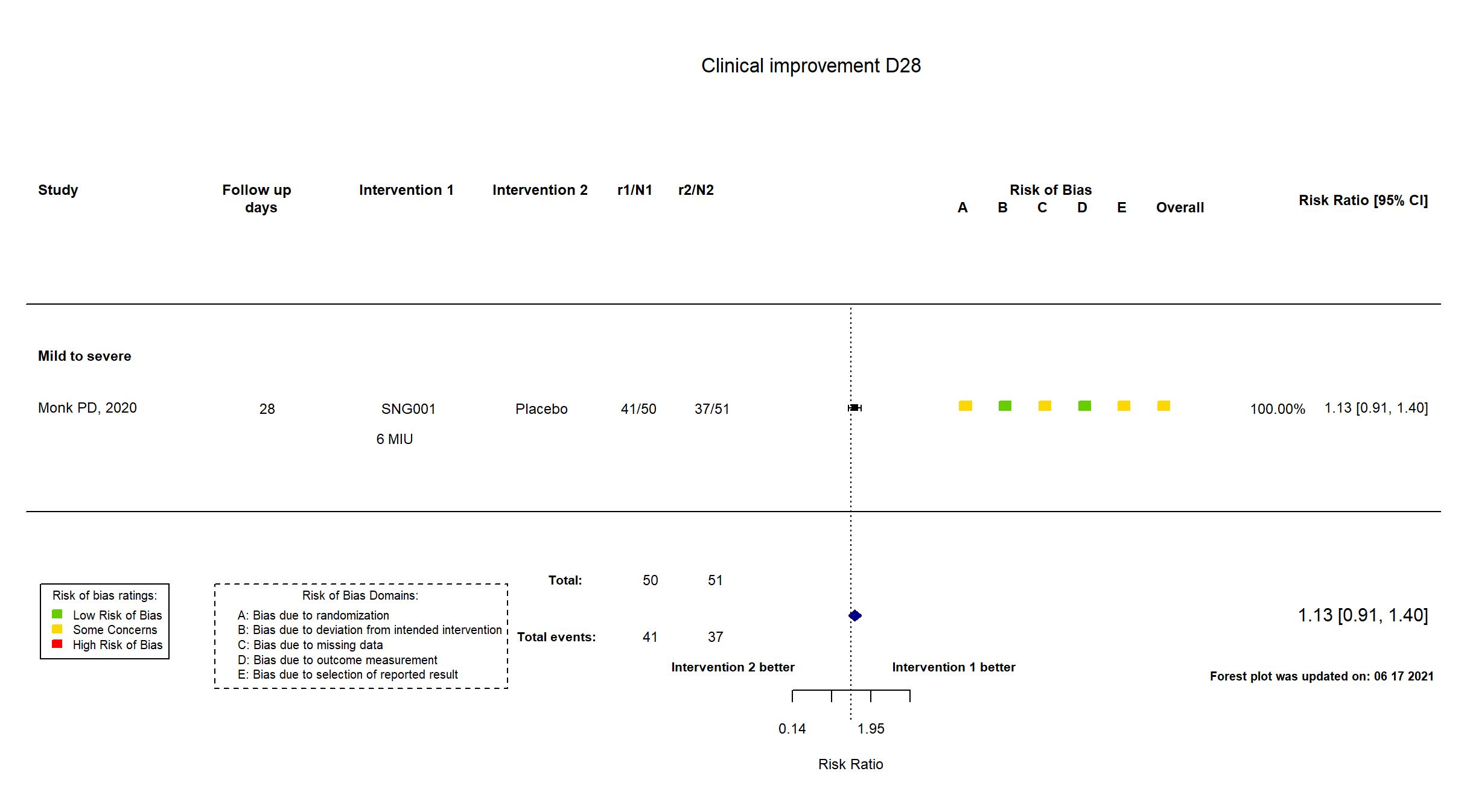






Trial NCT04385095 ; EudraCT 2020-001023-14
Publication Monk PD, Lancet Respir Med (2020) (published paper)
Dates: 2020-03-30 to 2020-05-30
Funding: Private (Synairgen Research, UK)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
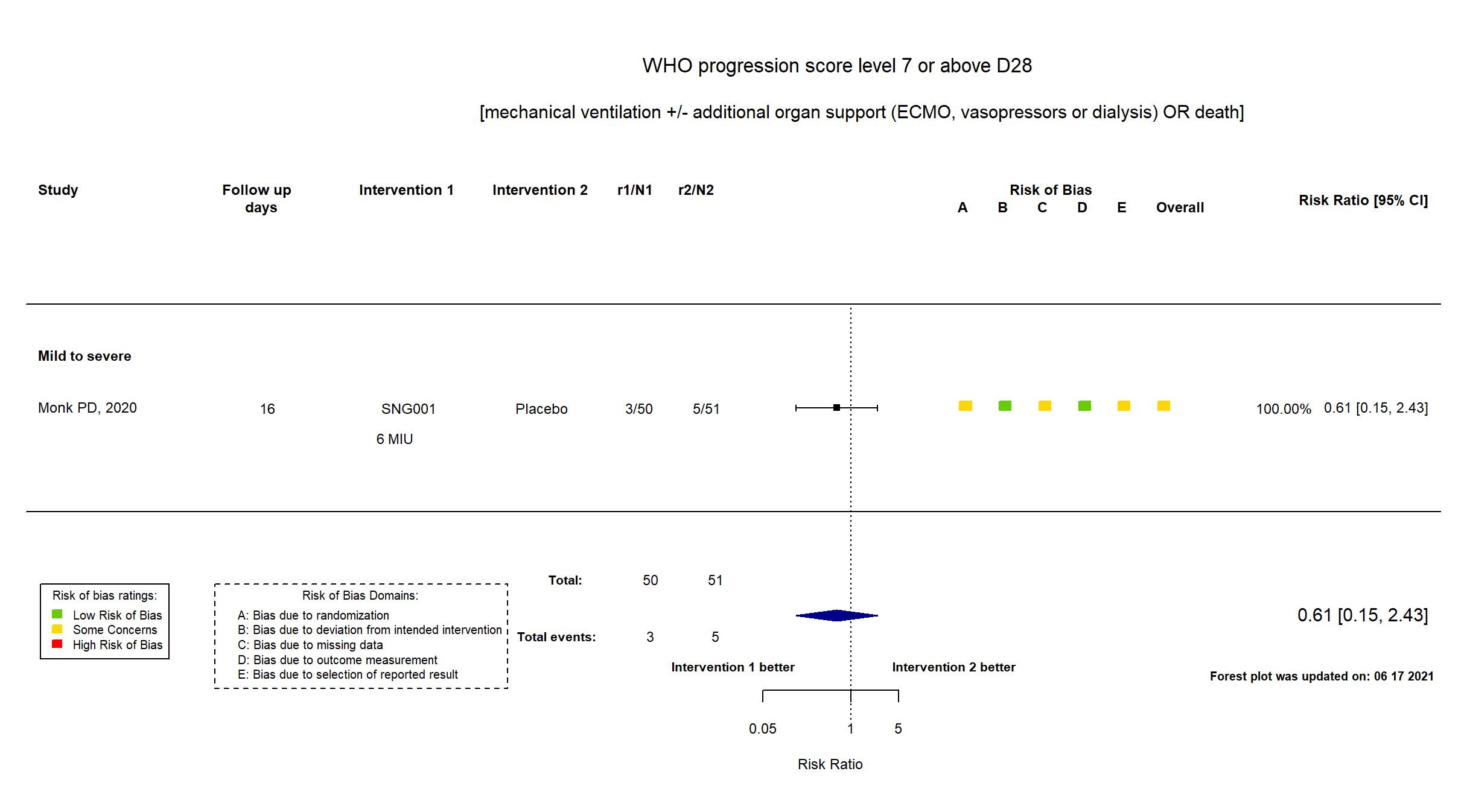
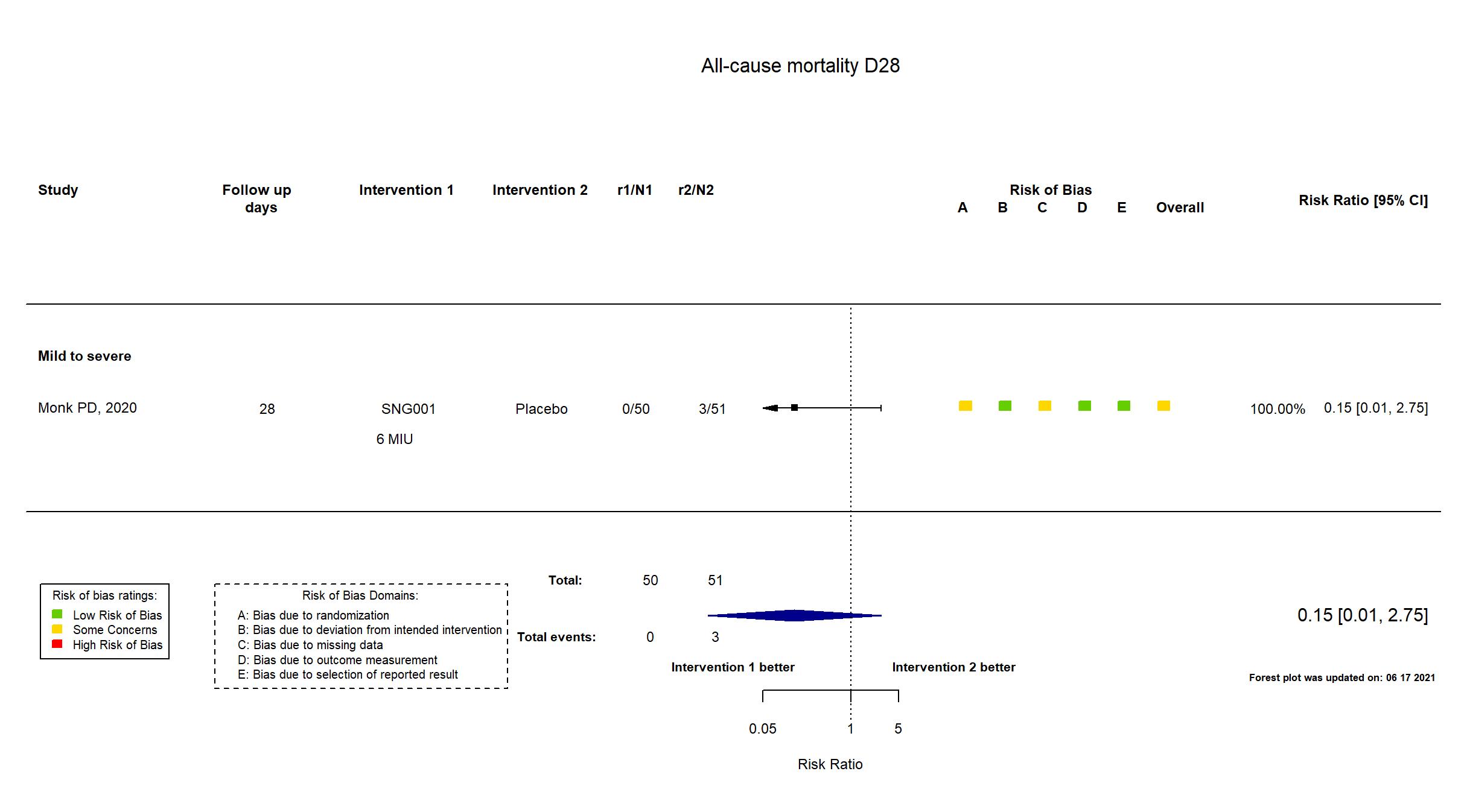
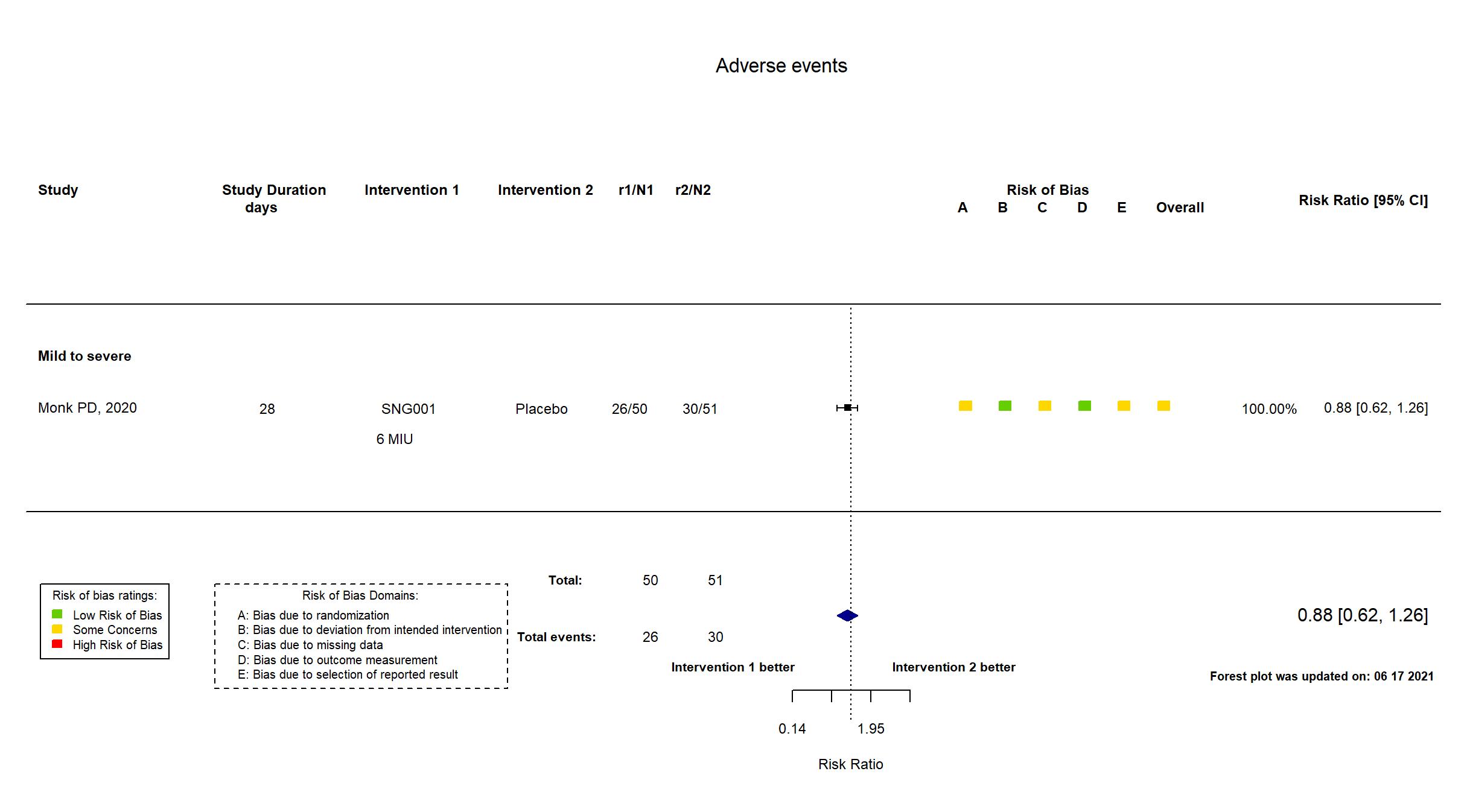
Multicenter / UK Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
SNG001 6 MIU delivered via nebuliser once a day for 14 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : SNG001 =50 Placebo=51 | |
| Characteristics of participants N= 101 Mean age : NR 58 males Severity : Mild: n=30 / Moderate: n=64 / Severe: n=2 Critical: n=0 | |
| Primary outcome | |
| In the register Change in condition measured using the Ordinal Scale for Clinical Improvement during the dosing period [ Time Frame: Day 1 to Days 15 and 28 ] | |
| In the report change in clinical condition on the WHO Ordinal Scale for Clinical Improvement (OSCI) during the dosing period | |
| Documents avalaible |
Protocol Yes. In English Statistical plan NR Data-sharing willing stated in the publication:
|
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the published article, the study registry and protocol were used in data extraction and risk of bias assessment. The study reached planned sample size and reports on the hospital setting part of the pilot phase. The home based setting part of the pilot phase had not been reported at the time of assessing this study. A pivotal phase was also planned. Several outcomes specified in the trial registry and protocol were not reported in the published article, including progression and evolution of pneumonia, National Early Warning Score 2 (NEWS2) assessment of acute-illness severity, concomitant medications, and virus clearance. There is no change from the trial registration in the intervention and control treatments. |