Ivermectin+Doxycycline vs Standard care/Placebo (RCT)
Hospitalized patients
FOREST PLOTS -2021-07-22







Trial NCT04407130
Publication Ahmed S, Int J Infect Dis (2020) (published paper)
Funding: Private (Beximco Pharmaceutical Limited, Bangladesh)
Conflict of interest: No
| Methods | |
| RCT Blinding: double blinding | |
| Location :
* / Bangladesh Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
IVM+DX Ivermectin : 12mg once-off orally. Doxycycline : 200mg orally at day-1 followed by 100mg twice a day for next 4 days Ivermectin 12mg once a day orally for 5 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : IVM+DX=24 Ivermectin=24 Placebo=24 | |
| Characteristics of participants N= 72 Mean age : NR 33 males Severity : Mild: n=72 / Moderate: n=0 / Severe: n=0 Critical: n=0 | |
| Primary outcome | |
| In the register 1.Virological clearance [ Time Frame: within 7 days after enrollment ] 2.Remission of fever [ Time Frame: within 7 days after enrollment ] 3.Remission of cough [ Time Frame: within 7 days after enrollment ] | |
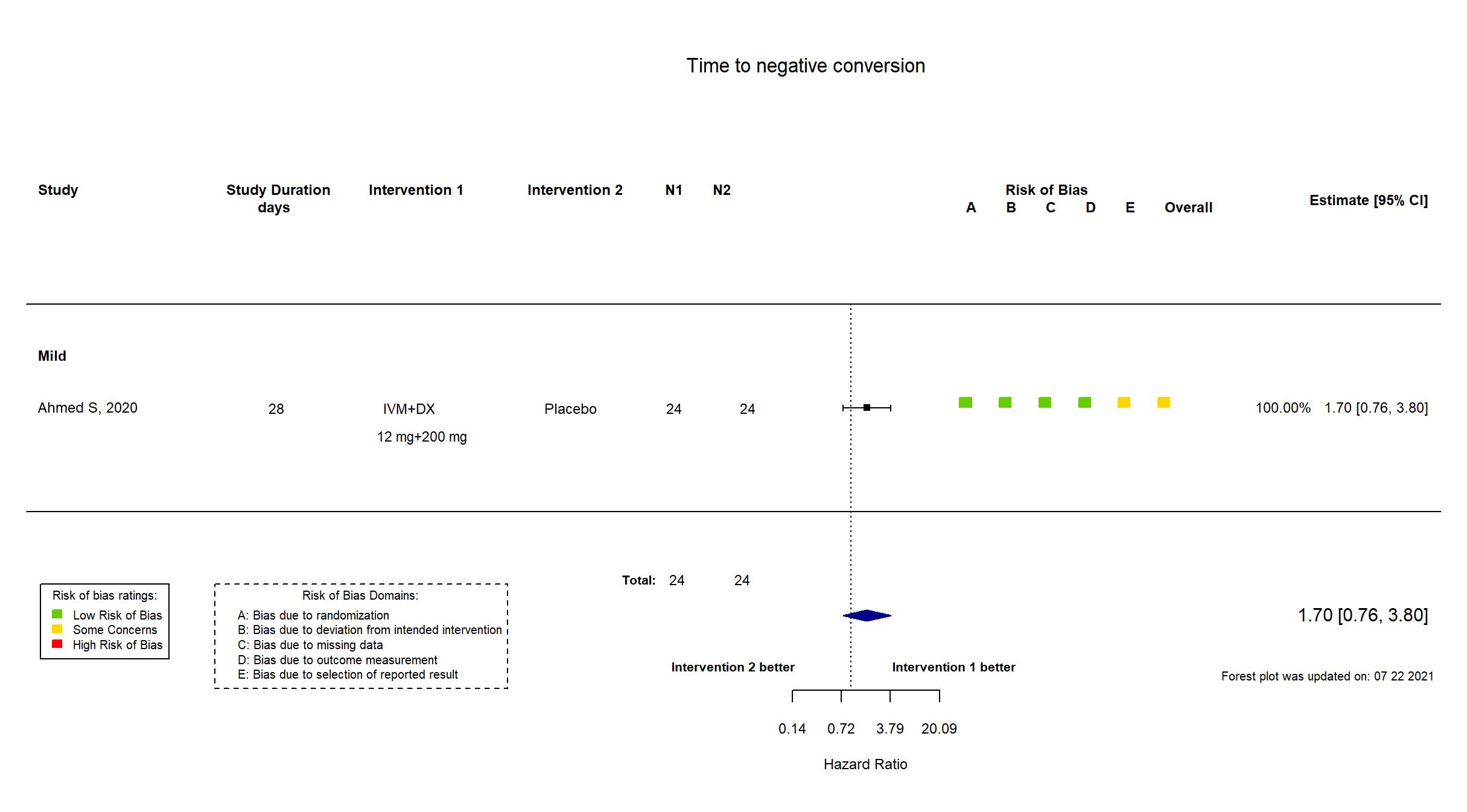
| In the report Time required for virological clearance (a negative rRT-PCR result on nasopharyngeal swab); remission of fever (≥37.5C) and cough within 7 days | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
The published article and the regsitry were used in data extraction and assessment of risk of bias. No study protocol or statistical analysis plan were available. 4/72 participants withdrew consent because of family obligations and unwillingness to be tested further. Baseline characteristics were not reported by study group. Some efficacy outcomes were not reported in the results section of the paper although they were listed in the methods section. Mortality, reported as a study outcome in the methods, was not clearly reported.
This study was updated on February 2nd, 2021 after contact with authors. |
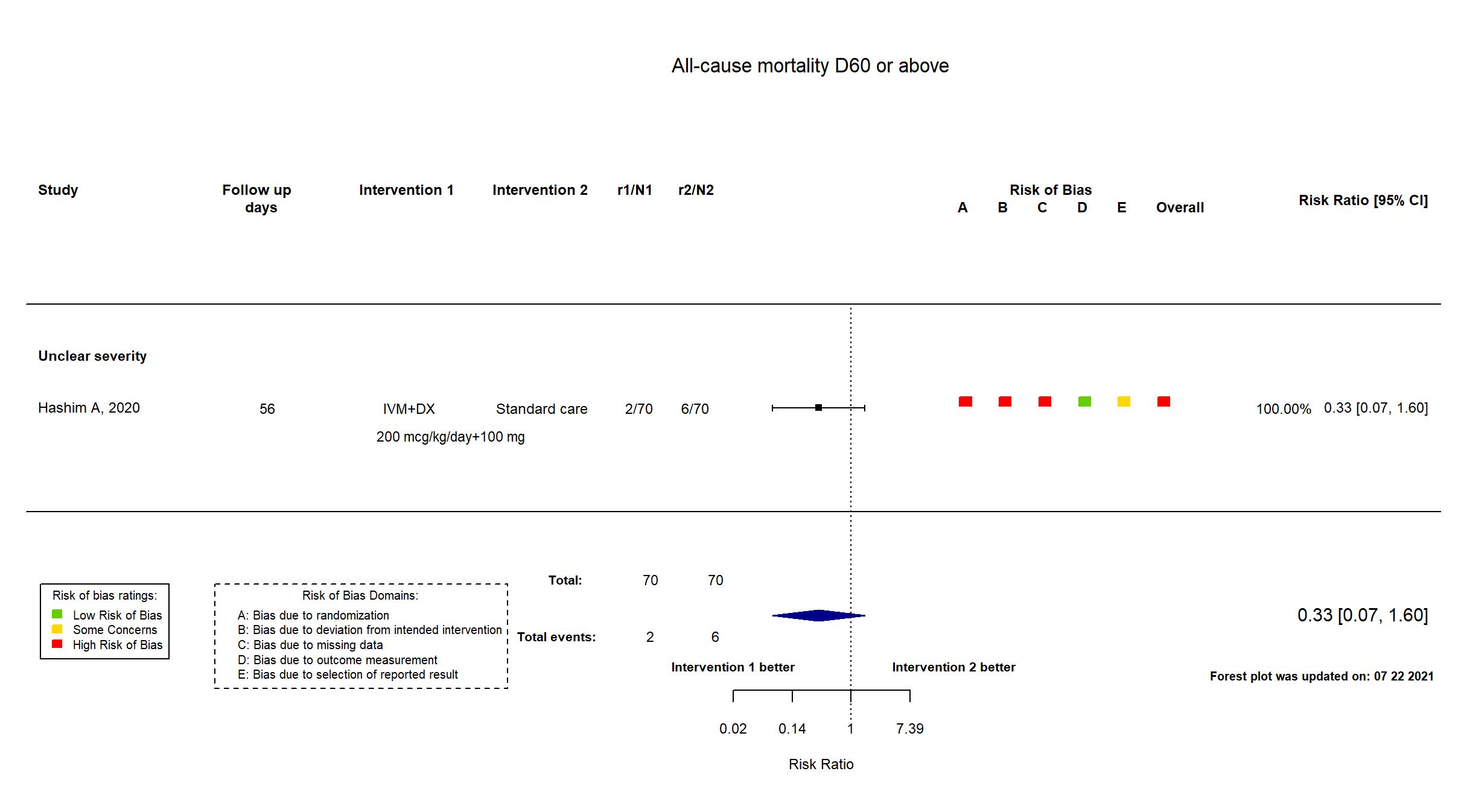
Trial NCT04591600
Publication Hashim A, medRxiv (2020) (preprint)
Dates: 2020-07-01 to 2020-09-30
Funding: Public/non profit (Baghdad-Alkarkh General Directorate of Health in Bghadad, Iraq)
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / Iraq Follow-up duration (days): 56 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
IVM+DX IVM: 200 mcg/kg/day PO for 2 days, third dose 7 days after the first, as needed; DX: 100 mg PO every 12 hrs for 5-10 days |
|
| Control
Standard care | |
| Participants | |
| Randomized NR Analyzed 140 participants IVM+DX=70 Standard care=70 | |
| Characteristics of participants N= 140 Mean age : NR 73 males Severity : Mild: n=* / Moderate: n=* / Severe: n=* Critical: n=* | |
| Primary outcome | |
| In the register 1. Mortality rate [ Time Frame: Up to 8 weeks ] The effect of the experimental drugs to reduce the mortality rate (death rate) of treated patients. 2. Rate of progression disease [ Time Frame: up to 8 weeks ] Rate of patients under treatment who undergo progression of disease to a more advanced stage | |
| In the report NR | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
High |
| General comment | In addition to all available versions of the pre-print, the study register was used in data extraction and risk of bias assessment. No sample size calculation was reported. The study used quasi-randomized allocation, with participants allocated to treatments according to their day of recruitment; this method was compromised in that all critical patients were allocated to the experimental treatment arm, reported as being for ethical reasons. The type of blinding extracted for the study was based on the registry as no information was stated in the report regarding this. There was also no primary or secondary outcomes reported in the paper, nor a flow chart or explicit mention of the absolute number of patients randomized per arm. |
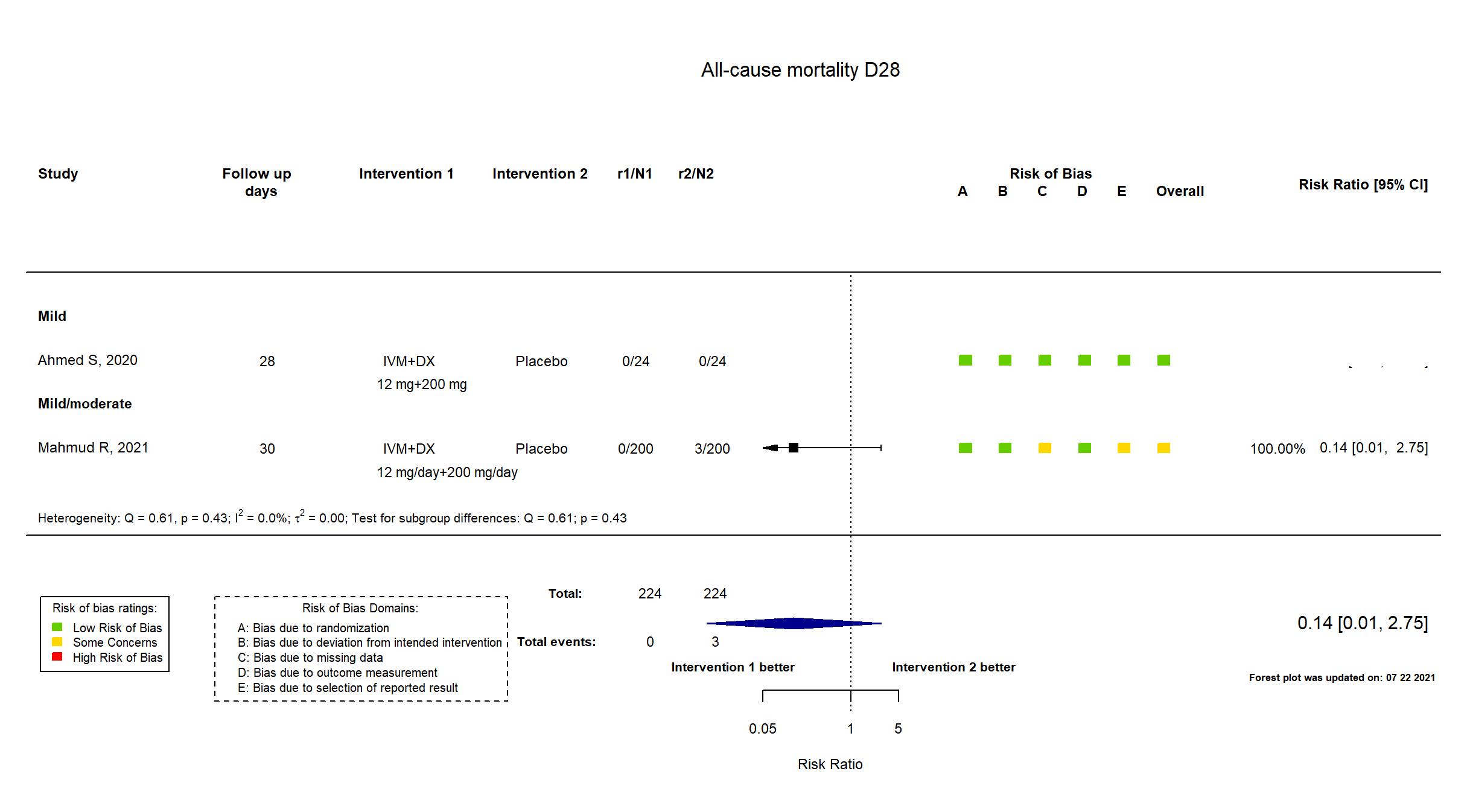
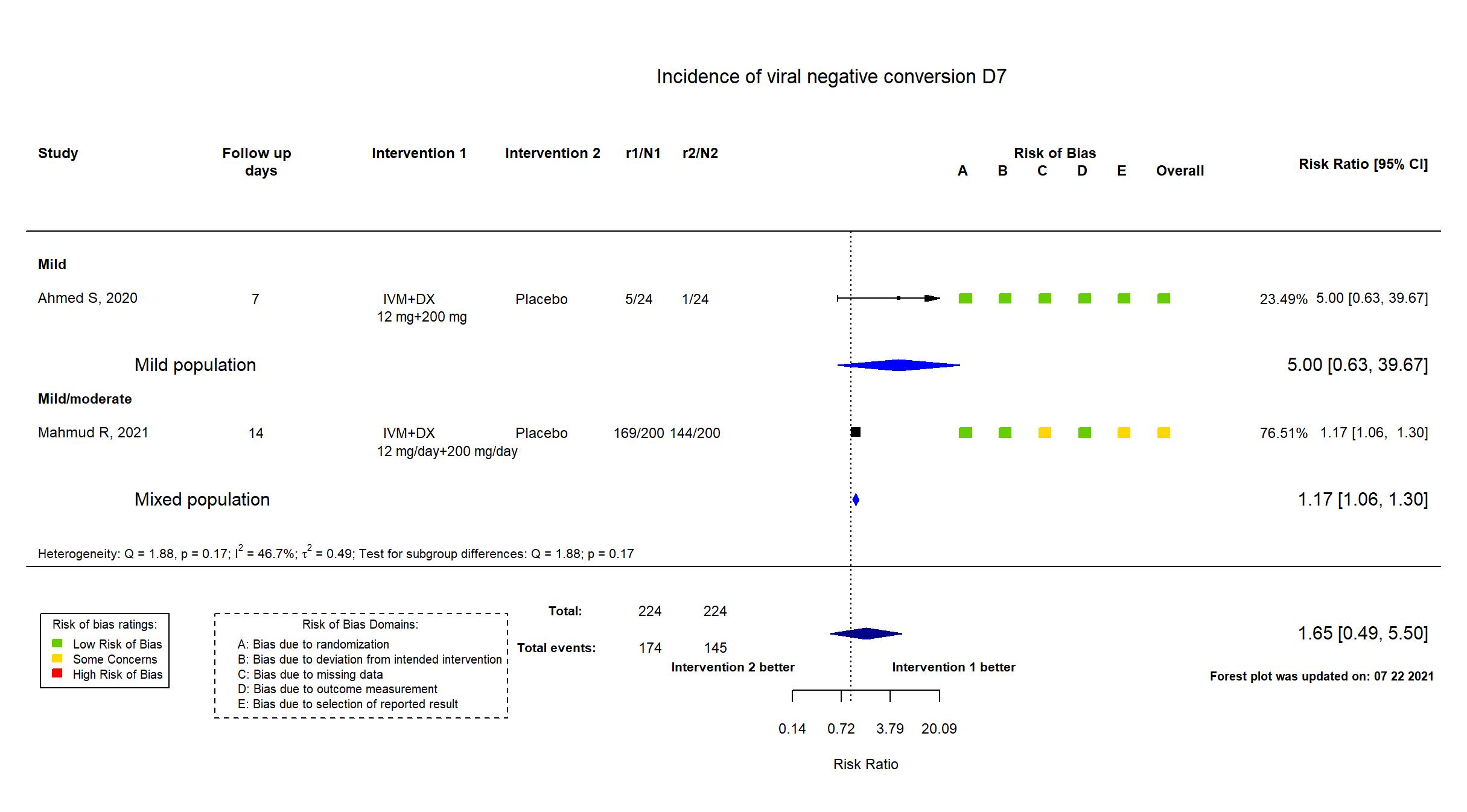
Trial NCT04523831
Publication Mahmud R, J Int Med Res (2021) (published paper)
Dates: 2020-06-01 to 2020-08-30
Funding: drug donation only (No specific grant; "Popular Pharmaceuticals Bangladesh Limited for supplying the study intervention and personal protective equipment")
Conflict of interest: No
| Methods | |
| RCT Blinding: | |
| Location :
Single center / Bangladesh Follow-up duration (days): 30 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
IVM+DX Ivermectin : 12 mg orally once off. Doxycycline : 100 mg orally twice a day for 5 days. |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : IVM+DX =200 Placebo=200 | |
| Characteristics of participants N= 400 Mean age : NR 235 males Severity : Mild: n=* / Moderate: n=* / Severe: n=* Critical: n=* | |
| Primary outcome | |
| In the register Number of Patients With Early Clinical Improvement [ Time Frame: 7 days ]; Number of Participants With Late Clinical Recovery [ Time Frame: 12 days ] | |
| In the report Number of days required for clinical recovery from day 1 | |
| Documents avalaible |
Protocol Yes. In English Statistical plan Yes Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
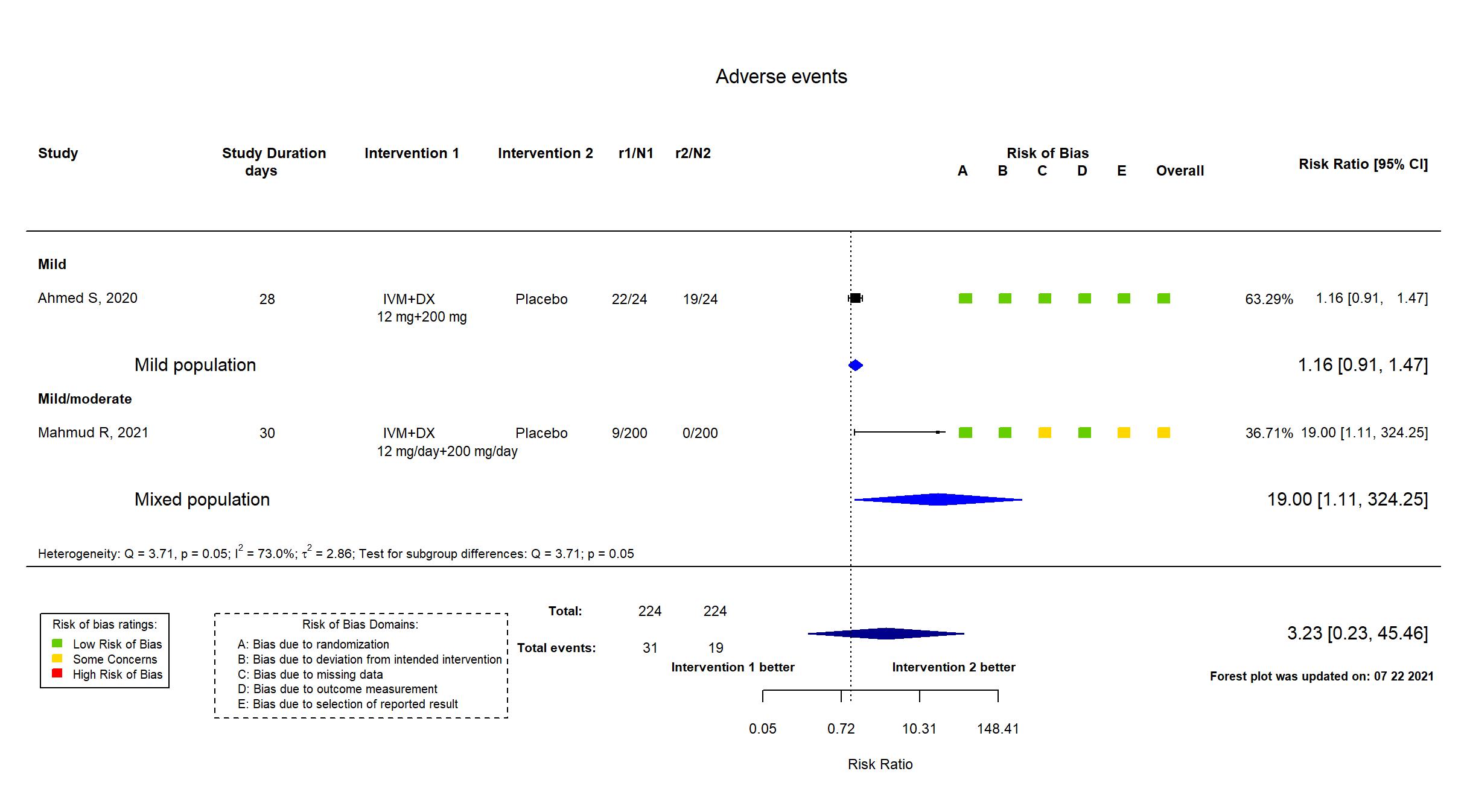
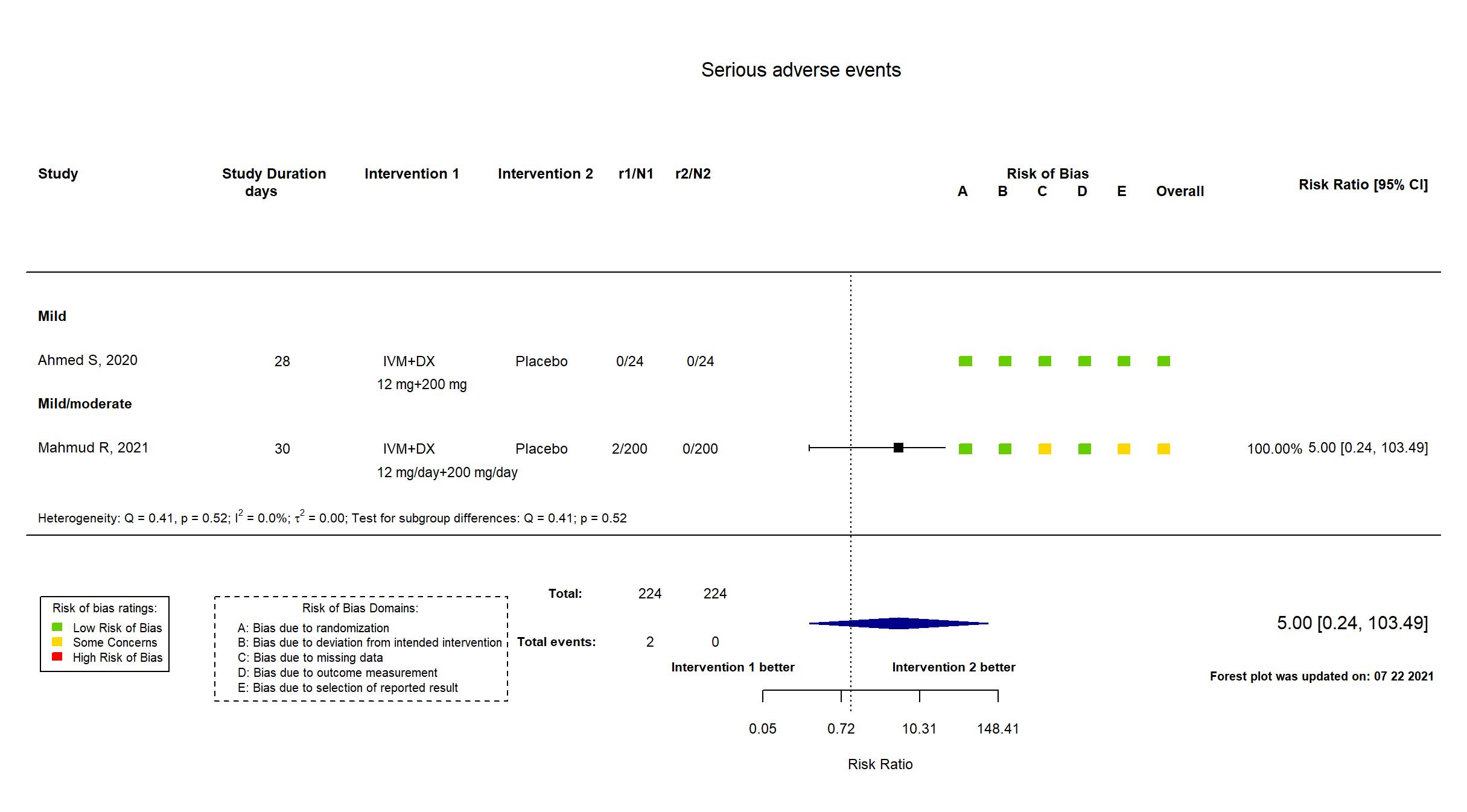
In addition to the published article, the study registry (retrospective), protocol and statistical analysis plan (retrospective) was used in data extraction and risk of bias assessment. Outcomes for adverse and serious adverse events were extracted from the result section of the study registry.Target sample size specified in the registry and protocol was achieved. There is no change from the trial registration or protocol in the intervention and control treatments or in the outcomes. However, the study registry and protocol were retrospective and therefore interpreted with caution. Denominators for SAEs/withdrawal due to AEs and Mortality do not seem to include the participants with these outcomes.
On 19th of May, 2021, this study was updated based on the published report. |