Nitazoxanide vs Standard care/Placebo (RCT)
Hospitalized patients
FOREST PLOTS -2022-01-04




Trial NCT04348409
Publication Blum VF, EClinicalMedicine (2021) (published paper)
Dates: 2020-05-20 to 2020-09-21
Funding: Private (Farmoquimica (FQM), Brazil)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: double blinding | |
| Location :
Multicenter / Brazil Follow-up duration (days): 21 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Nitazoxanide 600 mg orally twice a day for 7 days |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Nitazoxanide =25 Placebo=25 | |
| Characteristics of participants N= 50 Mean age : NR 15 males Severity : Mild: n=* / Moderate: n=* / Severe: n=0 Critical: n=0 | |
| Primary outcome | |
| In the register Viral load [ Time Frame: day 1, 4, 7, 14 and 21 ] | |
| In the report Virological response to treatment | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment |
In addition to the peer-reviewed journal publication and pre-print article, the trial registry was used in data extraction and assessment of risk of bias. Neither study protocol nor statistical analysis plan was available. The study achieved its pre-stated sample size. There is no significant difference from the trial registration in the population or intervention and control treatments. The scale used to categorize clinical status differed between the trial registry and the pre-print article, having been adapted to include fewer points for assessment of a small sample. The article reports adverse events and several laboratory outcomes that were not included in the registry. Only proportions were reported for clinical improvement (hospital discharge) with unclear denominators and number of cases. Viral negative conversion was reported, but at a follow-up duration that was too long for the COIVD-19 NMA (21 days); thus it was not included. On 26th of July, 2021, this study was updated based on updated pre-print.
On December 23rd, 2021, this study was updated after publication of a corrigendum. |
Trial NCT04463264
Publication Silva M, medRxiv (2021) (preprint)
Dates: 2020-07-01 to 2020-12-30
Funding: Private (Laboratorios Roemmers S.A.I.C.F.)
Conflict of interest: Yes
| Methods | |
| RCT Blinding: Single blinded, no restrictions | |
| Location :
Multicenter / Argentina Follow-up duration (days): 35 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Nitazoxanide 500 mg orally 4 times a day for 14 days. |
|
| Control
Placebo | |
| Participants | |
| Randomized participants : Nitazoxanide =33 Placebo=13 | |
| Characteristics of participants N= 46 Mean age : NR 26 males Severity : Mild: n=25 / Moderate: n=0 / Severe: n=0 Critical: n=0 | |
| Primary outcome | |
| In the register Eradication of SARS COV-2 from patients' respiratory tract secretions by treatment day 7th. [Time Frame: 7 day] | |
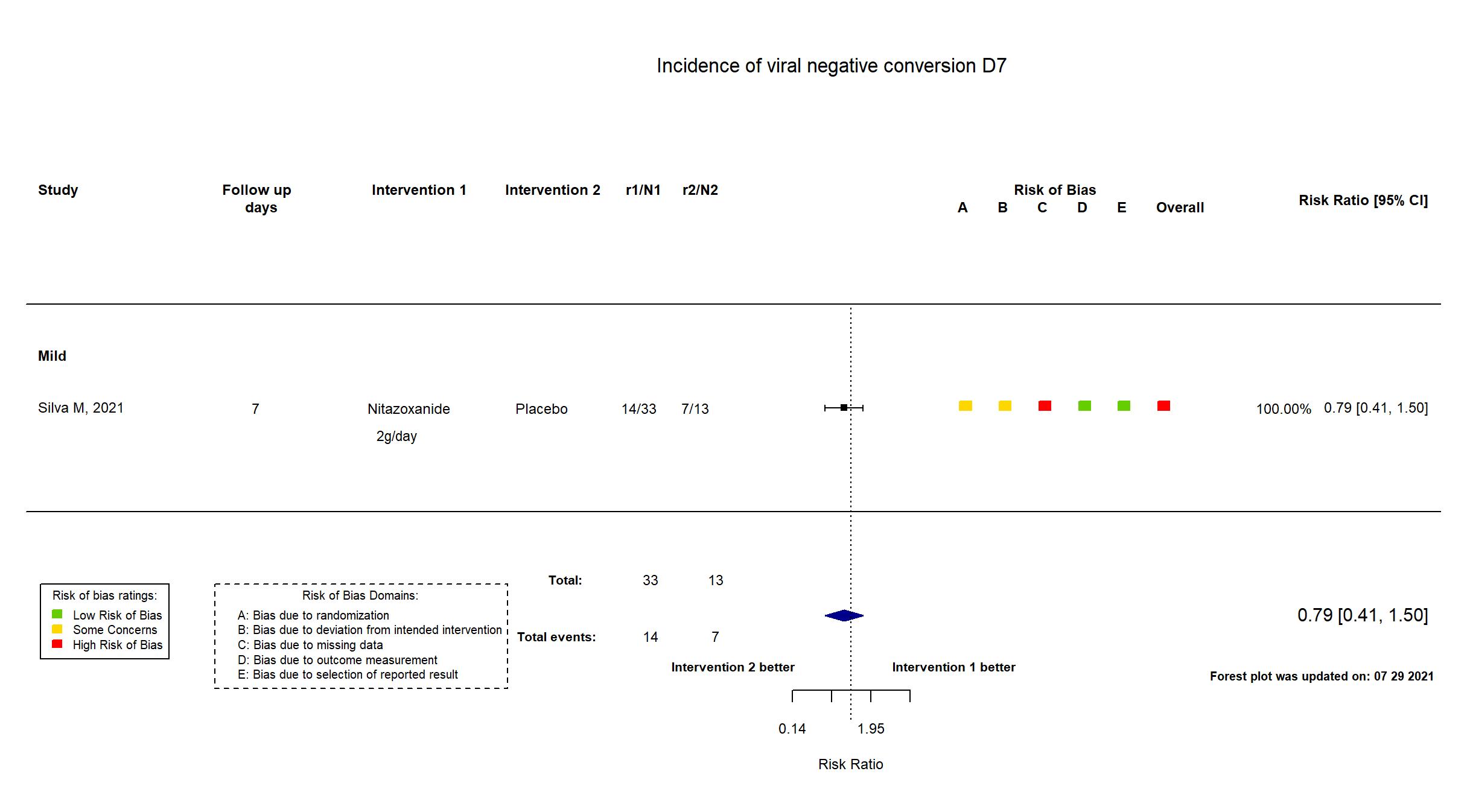
| In the report Viral eradication from the patients’ respiratory tract secretions on day 7 after starting treatment (a viral eradication of at least 35% would be clinically relevant). | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
High |
| General comment |
In addition to the available version of the pre-print article, the study registry was used in data extraction and risk of bias assessment. There is no change from the trial registration in the intervention and control treatments. The authors stated that the original protocol included an interim analysis to find any trends upon enrolment of 1/3 of the proposed number of patients (interim analysis). As a result, the target sample size specified in the registry was not achieved and some efficacy outcomes were not reported in the paper. In this interim analysis only virological outcomes are reported. Other outcomes in the trial registry such as clinical improvement (WHO scale), hospitalization and mechanical ventilation/ICU requirement are not reported. The primary and secondary efficacy analyses were performed as per protocol. It includes patients with complete data on days 1 and 7 and who received the indicated treatment (two patients without data on day 7 and one patient randomized to NTX but treated with doses well below the protocol doses were excluded). |