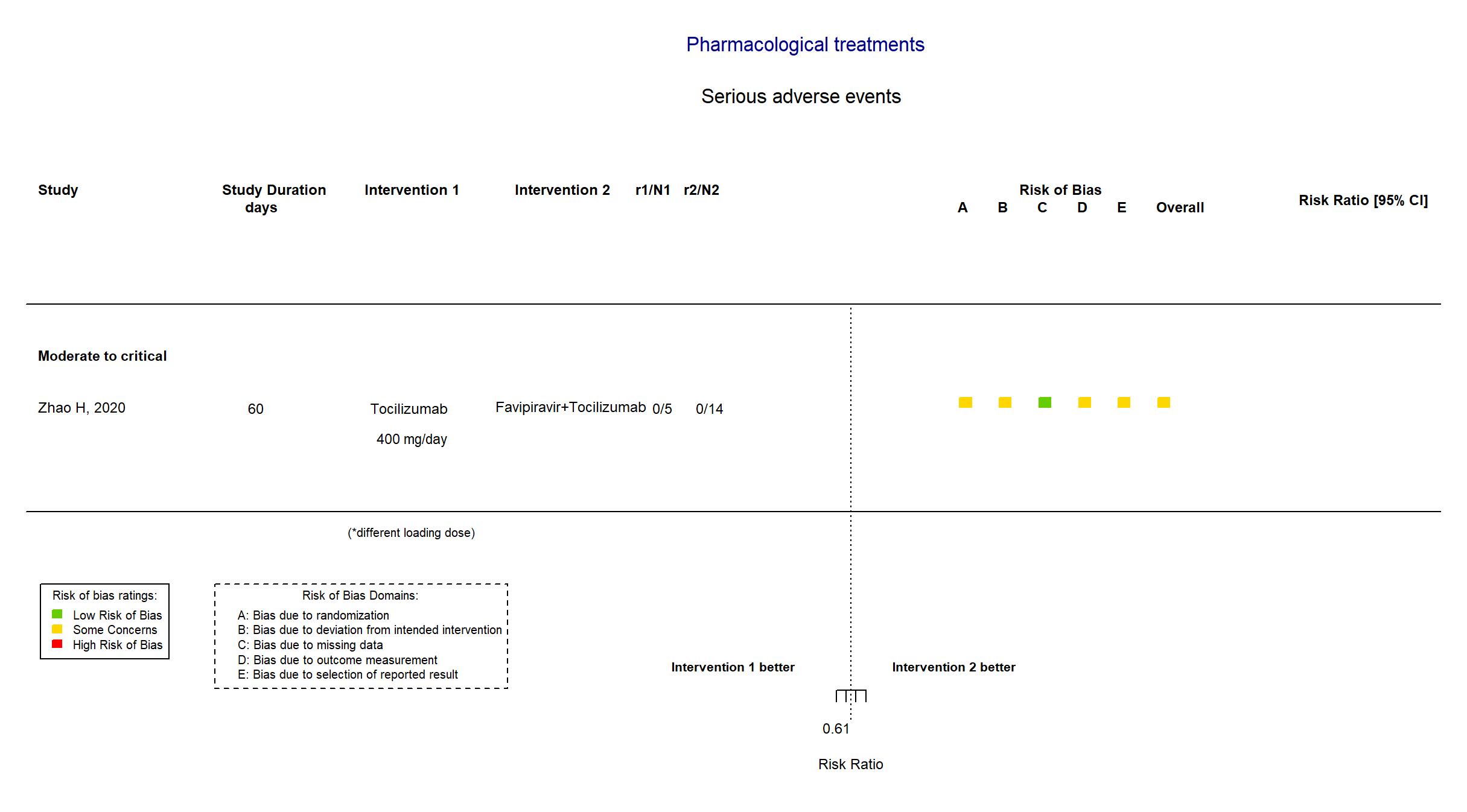
Tocilizumab vs Favipiravir+Tocilizumab (RCT)
Hospitalized patients
FOREST PLOTS -2021-08-13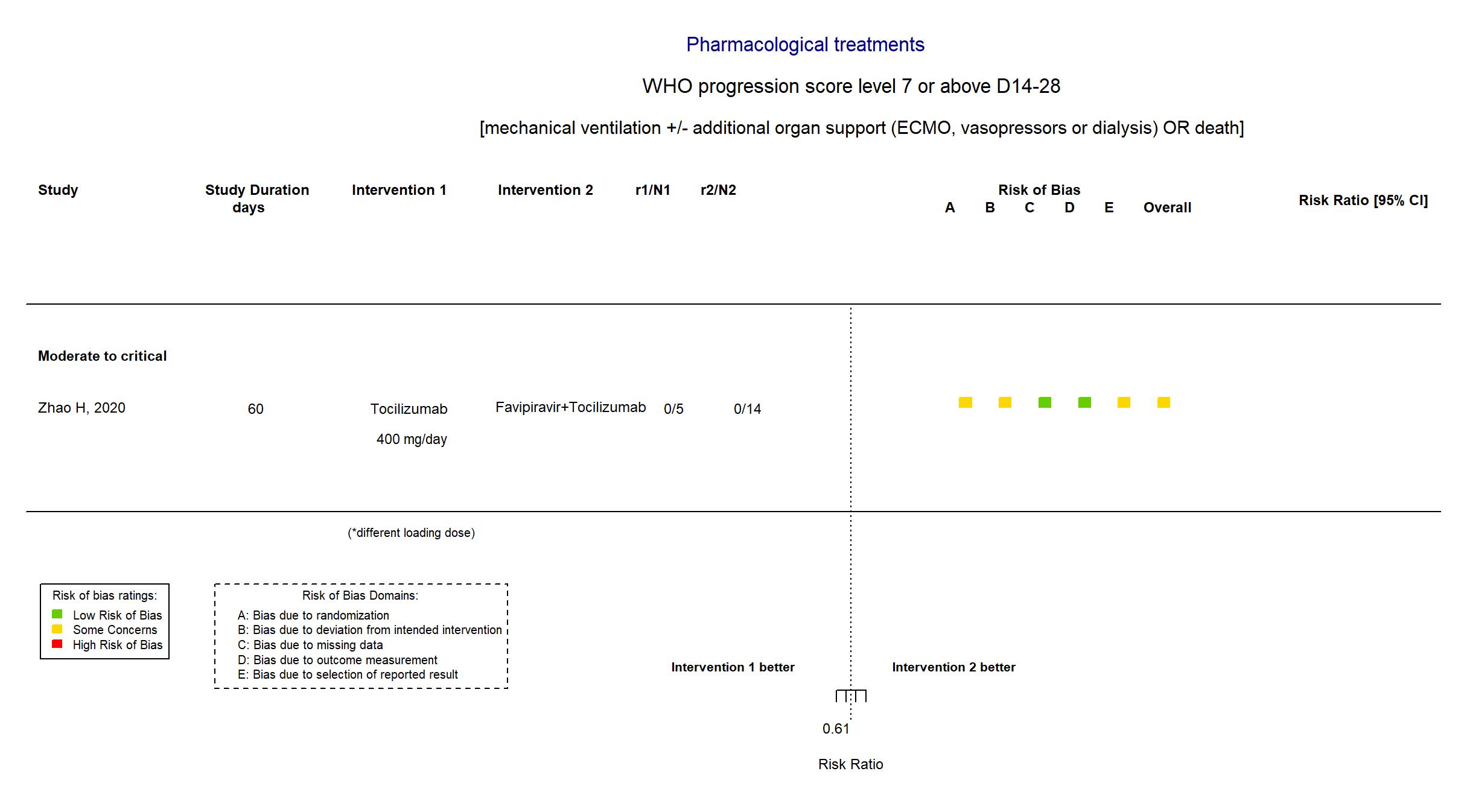


Trial NCT04310228
Publication Zhao H, Biomed Pharmacother (2020) (published paper)
Dates: 2020-02-02 to 2020-03-15
Funding: Public/non profit ( Chinese COVID-19 scientific research emergency project, China Mega-Project for Infectious Diseases, and China Mega-Project for Innovative Drugs)
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / China Follow-up duration (days): 60 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Favipiravir 1600 mg orally twice a day on the first day, followed by 600 mg orally twice a day for the next 6 days. Tocilizumab 4-8 mg/kg IV infusion (recommended dose: 400 mg), maximum 800 mg, a second dose could be administered 12 hours after the first. |
|
| Control
Favipiravir+Tocilizumab Favipiravir: 1600 mg orally twice a day on the first day, followed by 600 mg orally twice a day for the next 6 days. Tocilizumab: 4-8 mg/kg IV infusion (recommended dose: 400 mg), maximum 800 mg, a second dose could be administered 12 hours after the first. | |
| Participants | |
| Randomized participants : Favipiravir=7 Tocilizumab=5 Favipiravir+Tocilizumab=14 | |
| Characteristics of participants N= 26 Mean age : NR 14 males Severity : Mild: n=0 / Moderate: n=12 / Severe: n=13 Critical: n=1 | |
| Primary outcome | |
| In the register Clinical cure rate [Time Frame: 3 months ] Definition of clinical cure: The viral load of the respiratory specimen was negative for two consecutive times (the interval between the two tests was greater than or equal to one day), the lung image improv | |
| In the report cumulative lung lesion remission rate (lung CT (lung CTexamination indicated absorption of lung inflammation) | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Not reported |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
Some concerns |
| General comment | In addition to the available pre-proof published version of the article, the study registries were used in data extraction and risk of bias assessment. The protocol and the statistical analysis plan were not available. The target sample size of n=150 specified in the registry was not achieved (actual sample size n=26). There is no change from the trial registries in the intervention and control treatments. Some efficacy outcomes specified in the registries were not reported in the paper,and the primary outcome reported differs in the report and registries. Both adverse events and serious adverse events were not stated as outcomes in the trial registries. |