Inhaled corticosteroids vs Standard care/Placebo (RCT)
Hospitalized patients
Studies included but not extracted/included in the analysis: Terada J, Drug Discov Ther, 2022
FOREST PLOTS -2022-10-07
Studies included but not extracted/included in the analysis: Terada J, Drug Discov Ther, 2022
FOREST PLOTS -2022-10-07


Trial *
Publication Alsultan M, Interdiscip Perspect Infect Dis (2021) (published paper)
Dates: 2021-08-01 to 2021-08-30
Funding: Not reported/unclear
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Single center / Syria Follow-up duration (days): * | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Colchicine Initial dose : 1.5 mg orally followed by 0.5 mg 1 hour later on day 1, Maintenance dose: 0.5 mg orally twice daily for 4 days Budesonide 200 mcg inhaled twice daily for 5 days |
|
| Control
Standard care | |
| Participants | |
| Randomized participants : Colchicine=14 Budesonide =14 Standard care=21 | |
| Characteristics of participants N= 49 Mean age : NR 19 males Severity : Mild: n=0 / Moderate: n=* / Severe: n=* Critical: n=0 | |
| Primary outcome | |
| In the register NR | |
| In the report NR | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
* |
| General comment | Only the published article was available for data extraction and assessment of risk of bias. No registry, protocol or statistical analysis plan was available. No outcome was identified as primary in the article. It is not clear whether the study achieved a target sample size. No outcomes with clear time points relevant for COVID-NMA were reported. |
Trial NCT04330586
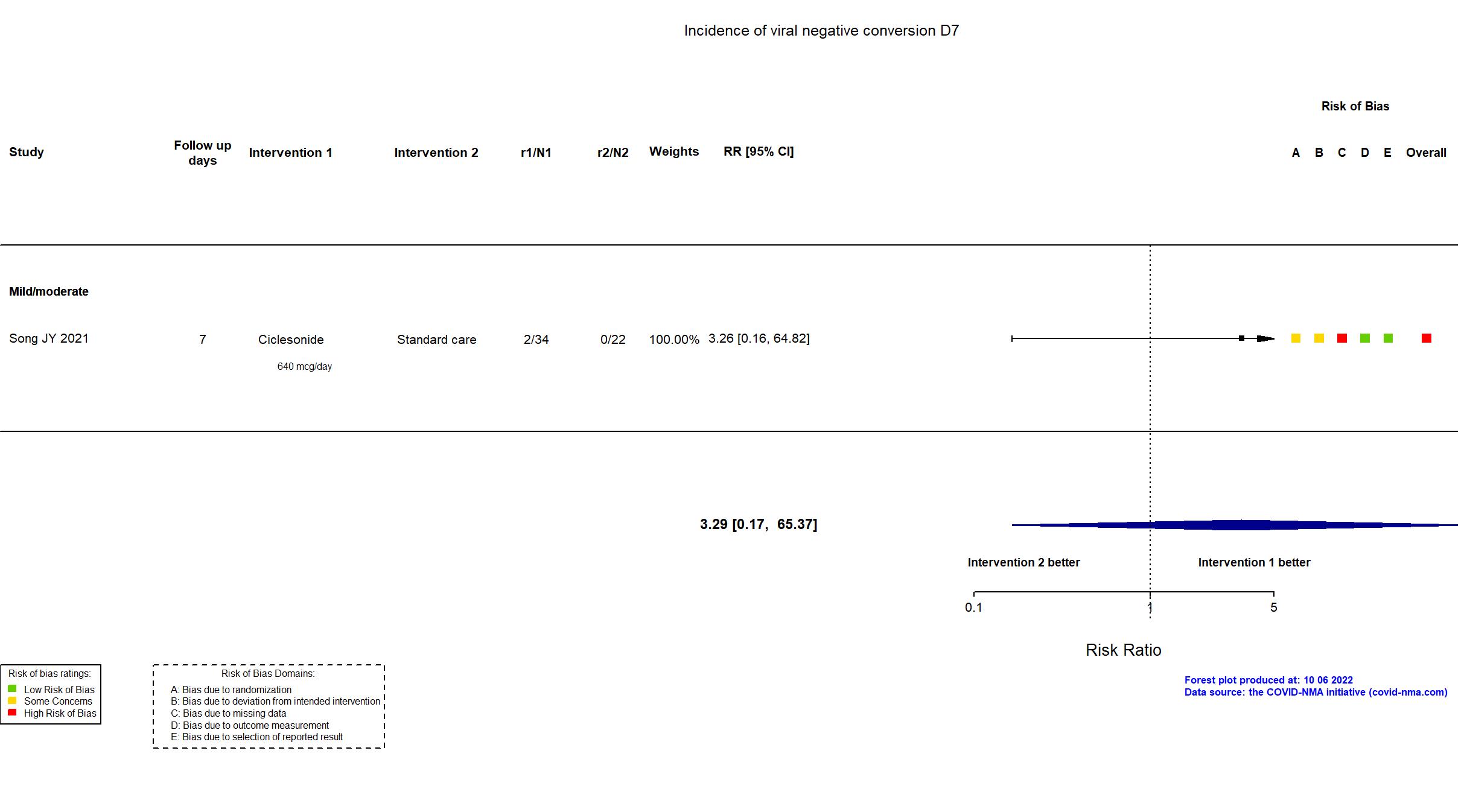
Publication Song JY, J Clin Med (2021) (published paper)
Dates: 2020-05-08 to 2021-03-31
Funding: Mixed (National Research Foundation of Korea; Korea University Guro Hospital grant that was underwritten by the SK Chemicals Corporation)
Conflict of interest: No
| Methods | |
| RCT Blinding: Unblinded | |
| Location :
Multicenter / South Korea Follow-up duration (days): 28 | |
| Inclusion criteria |
|
| Exclusion criteria |
|
| Interventions | |
| Treatment
Ciclesonide 320 mcg inhaled twice daily for 14 days |
|
| Control
Standard care | |
| Participants | |
| Randomized NR Analyzed 61 participants Ciclesonide=35 Standard care=26 | |
| Characteristics of participants N= 61 Mean age : NR 20 males Severity : Mild: n=* / Moderate: n=* / Severe: n=0 Critical: n=0 | |
| Primary outcome | |
| In the register Rate of SARS-CoV-2 eradication at day 14 from study enrollment [ Time Frame: Hospital day 14 ] Viral load | |
| In the report SARS-CoV-2 eradication rate based on qRT-PCR on day 14 of study enrollment | |
| Documents avalaible |
Protocol NR Statistical plan NR Data-sharing willing stated in the publication: Yes |
| Risk of bias Overall The overall risk of bias reported in the table corresponds to the highest risk of bias for the outcomes assessed for the systematic review |
High |
| General comment | In addition to the published article, the prospective registry was used in data extraction and assessment of risk of bias. Neither protocol not statistical analysis plan was available. The primary outcome reported reflects that in the registry. There were no substantive differences between the registry and the article in outcomes. During the trial period, evidence of the ineffectiveness of hydroxychloroquine was published and a third ciclesonide plus hydroxychloroquine arm was combined with the ciclesonide alone arm. Eight patients in the ciclesonide group received oral HCQ treatment concomitantly for 10 days. 68 participants were randomized and only 61 were analyzed. Reasons for exclusion from analysis are reported but arms are not indicated. |